High Performance Medical Plastic Market Size, Share, and Growth Forecast 2026 - 2033
Key Market Highlights
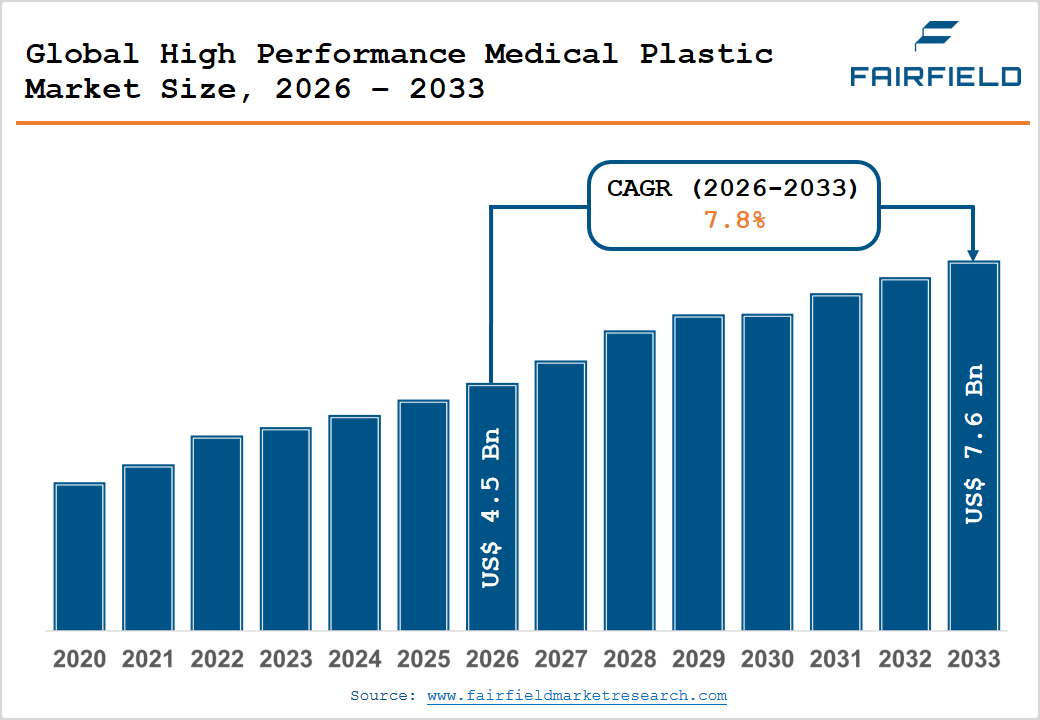
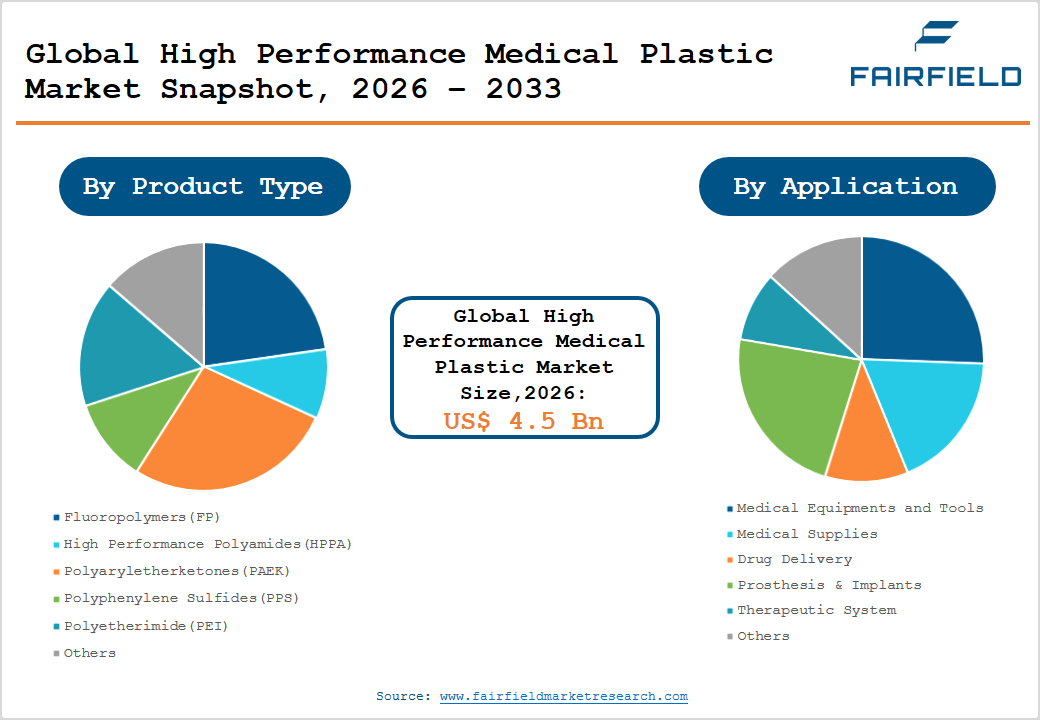
- The global High Performance Medical Plastic Market size is likely to be valued at US$ 4.5 Billion in 2026 and is expected to reach US$ 7.6 Billion by 2033, growing at a CAGR of 7.80% during the forecast period from 2026 to 2033.
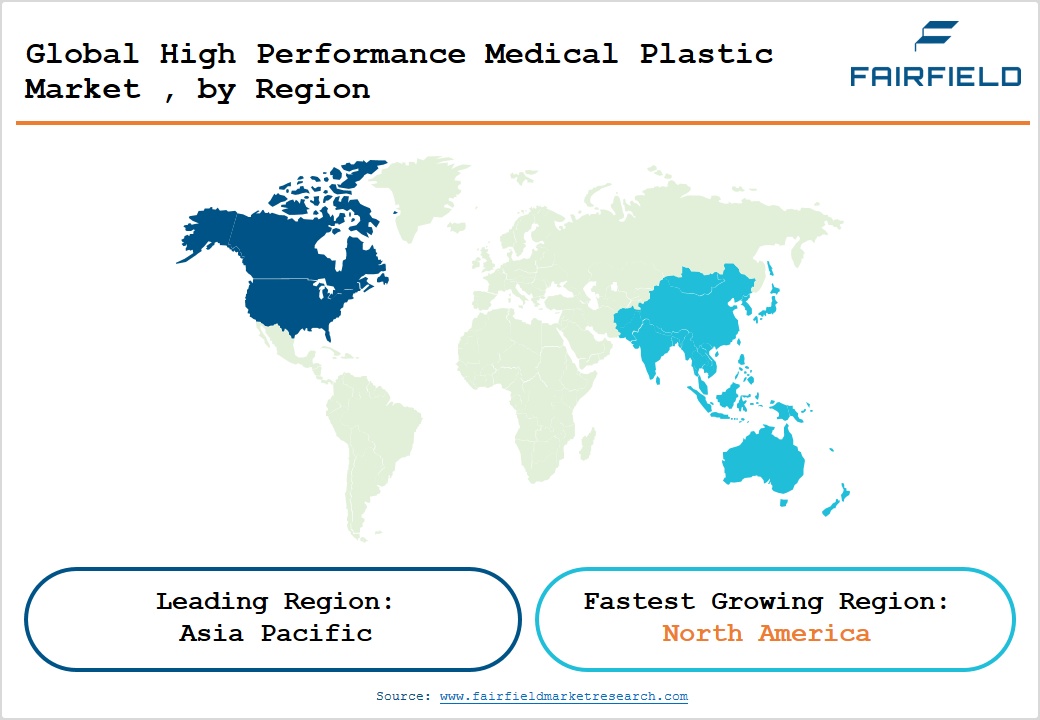
- Leading Region Market: Asia Pacific dominates the High Performance Medical Plastic market with approximately 59% share in 2026, driven by China's expansive medical device manufacturing base, India's growing healthcare infrastructure, and ASEAN's cost-competitive production environments.
- Fastest Growing Region Market: North America is the fastest growing regional market, fuelled by strong FDA regulatory frameworks, rising robotic-assisted surgery adoption, and multi-billion-dollar healthcare infrastructure spending commitments through 2028.
- Dominant Product Segment: Fluoropolymers lead the Product Type category with approximately 29% market share in 2026, owing to superior chemical inertness, biocompatibility, and compliance with FDA 21 CFR and ISO 10993 standards in medical tubing and catheter applications.
- Fastest Growing Application Segment: Prosthesis & Implants is the fastest growing application segment, driven by surging demand for PEEK-based orthopedic and spinal implants as aging populations expand globally, particularly in North America and Europe.
- Key Market Opportunity: Advanced drug delivery and bioresorbable implant technologies represent the most significant growth opportunity, supported by NIH funding exceeding US$ 1.5 billion and growing oncology and chronic disease treatment demand worldwide.
Market Dynamics
Market Growth Drivers
- Surging Demand for Biocompatible Materials in Medical Devices
The increasing complexity of medical devices has accelerated the adoption of high-performance medical plastics such as Polyaryletherketones (PAEK) and Polyetherimide (PEI), renowned for their exceptional biocompatibility, sterilizability, and dimensional stability. According to the U.S. Food and Drug Administration (FDA), the number of premarket approvals for implantable medical devices incorporating advanced polymers has risen steadily in recent years, reflecting a broader industry pivot toward polymer-first design strategies. Additionally, the global medical device market surpassed US$ 500 billion in 2023, per OECD Health Statistics, creating a robust downstream pull for specialty polymers. Enhanced mechanical strength, chemical inertness, and radio-transparency of high-performance plastics make them indispensable for orthopedic implants, dental prosthetics, and minimally invasive surgical tools.
- Rising Geriatric Population and Chronic Disease Burden
The global aging demographic is a key structural tailwind for the high-performance medical plastic market. The United Nations Department of Economic and Social Affairs (UN DESA) projects that the global population aged 65 years and above will reach 1.6 billion by 2050, approximately double the current figure. This translates directly into elevated demand for orthopedic devices, cardiovascular implants, and drug delivery systems, all of which are increasingly manufactured using high-performance polymers such as Polyphenylene Sulfide (PPS) and High Performance Polyamides (HPPA). The World Heart Federation reports that cardiovascular diseases claim approximately 17.9 million lives annually, intensifying demand for polymer-based therapeutic systems and diagnostics.
Market Restraints
- High Material and Processing Costs
A fundamental challenge constraining widespread adoption of high-performance medical plastics is their elevated production cost relative to commodity thermoplastics. Advanced engineering polymers such as PEEK (Polyetheretherketone) command prices exceeding US$ 80-100 per kilogram several times higher than general-purpose materials owing to complex multi-step synthesis routes and stringent purity standards. For smaller contract manufacturers and cost-sensitive healthcare markets, such pricing creates a significant adoption barrier, often limiting usage to premium or high-acuity applications and curbing broader penetration across the medical device supply chain.
- Stringent Regulatory Compliance and Extended Approval Timelines
The rigorous regulatory ecosystem governing medical-grade materials represents a persistent market restraint. In the United States, the FDA's 21 CFR Part 820 quality system regulations, and in Europe, the EU Medical Device Regulation (MDR 2017/745), mandate extensive biocompatibility and cytotoxicity testing per ISO 10993 standards. The average pre-market approval cycle for implantable polymer-based devices can extend 3-7 years, raising development costs and delaying time-to-market. This regulatory burden disproportionately affects mid-sized suppliers seeking to commercialize novel high-performance polymer formulations.
Market Opportunities
- Expansion of Minimally Invasive Surgery (MIS) and Robotic-Assisted Procedures
The rapid proliferation of minimally invasive and robotic-assisted surgical platforms presents a high-value opportunity for high-performance medical plastic suppliers. According to the American Hospital Association (AHA), over 70% of U.S. hospitals have invested in robotic surgical systems, driving demand for polymer components that offer precise tolerances, radiolucency, and MRI compatibility. Fluoropolymers (FP) and PEI are increasingly specified in endoscopic sheaths, laparoscopic instrument handles, and catheter materials due to their low friction coefficient and thermal stability. Intuitive Surgical reported over 7,700 da Vinci systems installed globally as of 2023, each requiring specialized polymer-based components, underscoring significant supply chain expansion opportunities for manufacturers.
- Growth in Advanced Drug Delivery and Bioresorbable Implant Technologies
Bioresorbable and controlled drug delivery polymer technologies are emerging as a transformative frontier in medical plastics. The U.S. National Institutes of Health (NIH) has significantly increased funding for polymer-based drug delivery platforms, with grants exceeding US$ 1.5 billion directed toward controlled-release and targeted therapy research through 2025. High-performance polymers including PAEK-based compounds and specialty fluoropolymers are finding new application in implantable drug eluting stents, biodegradable orthopedic screws, and micro-dosing capsules. The global drug delivery device segment is expanding in parallel with oncology and chronic pain management therapies, providing manufacturers with differentiated revenue streams that are less susceptible to cost pressures affecting commodity medical plastics.
Segmental Insights
- Product Type Analysis
Among all product type segments, Fluoropolymers (FP) command the leading position in the High Performance Medical Plastic market, accounting for approximately 29% of the total market share in 2026. This dominance is attributable to fluoropolymers' unparalleled chemical inertness, extremely low friction coefficient, high thermal stability (withstanding temperatures up to 260°C), and superior electrical insulation properties all critical attributes in medical tubing, catheters, and minimally invasive surgical instruments. PTFE (polytetrafluoroethylene) and PVDF (polyvinylidene fluoride) variants are cleared under FDA 21 CFR for food and drug contact, and their compliance with ISO 10993 biocompatibility testing further underpins widespread adoption in both implantable and non-implantable medical applications globally.
- Application Analysis
The Medical Equipment and Tools segment is the leading application category, representing approximately 34% share in 2026. This segment's primacy is driven by the expansive use of high-performance plastics in diagnostic imaging components, surgical handles, endoscopy devices, and sterilizable lab equipment. The Association for the Advancement of Medical Instrumentation (AAMI) highlights that over 40% of modern surgical instrument components rely on engineering-grade polymers to achieve weight reduction, corrosion resistance, and autoclave compatibility. The shift toward single-use surgical instruments accelerated by infection control protocols following the COVID-19 pandemic has further expanded volumetric demand for high-performance polymers within this application segment.
Regional Insights
- North America High Performance Medical Plastic Trends
North America represents the fastest growing region in the global High Performance Medical Plastic market, propelled by a well-established medical device manufacturing ecosystem, strong FDA regulatory clarity for polymer-based devices, and robust healthcare expenditure. The U.S. Centers for Medicare & Medicaid Services (CMS) projects national health spending to reach US$ 6.2 trillion by 2028, reflecting the depth of demand for advanced medical materials. Moreover, U.S. medical device exports surpassed US$ 56 billion in 2022 per the U.S. International Trade Administration, reinforcing domestic production requirements.
Key innovation clusters in Minnesota (medical device corridor), California, and Massachusetts are driving R&D in implantable polymer solutions. Companies such as DuPont and Celanese Corporation are investing significantly in expanding domestic capacity for medical-grade polymers, aligning with the CHIPS and Science Act's broader mandate to re-shore advanced materials manufacturing.
- Europe High Performance Medical Plastic Trends
Europe maintains a significant presence in the High Performance Medical Plastic market, underpinned by strong device manufacturing industries in Germany, France, United Kingdom, and Switzerland. The implementation of the EU Medical Device Regulation (EU MDR 2017/745) has tightened material qualification requirements, creating favourable conditions for premium high-performance polymer suppliers. Germany, home to global polymer giants Evonik Industries AG and BASF SE, leads regional production, while France's Arkema SA has strengthened its high-performance polymer portfolio.
The European Commission's Horizon Europe program has committed over €95.5 billion to research and innovation through 2027, with healthcare materials among priority themes. Spain and Italy are emerging as growing markets as their national healthcare systems modernise surgical infrastructure, while the United Kingdom post-Brexit continues to align with UKCA marking requirements, driving compliance-led polymer procurement.
- Asia Pacific High Performance Medical Plastic Trends
Asia Pacific is the leading region in the High Performance Medical Plastic market, commanding approximately 59% market share in 2026. China is the region's dominant force, supported by the National Medical Products Administration (NMPA)'s streamlined approval pathways for domestically produced medical devices and a vast manufacturing base. Japan contributes through precision medical device engineering, while India's National Health Mission and a rapidly growing medical devices sector targeted to reach US$ 50 billion by 2030 per the Government of India Ministry of Chemicals and Fertilizers create significant local demand.
ASEAN nations, particularly Malaysia, Thailand, and Vietnam, are attracting medical device manufacturing foreign direct investment due to competitive labour costs and improving regulatory infrastructure. Mitsubishi Chemical Corp and Solvay SA have expanded their regional polymer supply networks to serve these growth markets, reinforcing Asia Pacific's position as both the largest consumer and fastest-growing manufacturer of high-performance medical plastics.
Competitive Landscape
The High Performance Medical Plastic market exhibits a moderately consolidated structure, with the top 10 players collectively accounting for over 55% of the global market in 2026. Leading corporations such as Solvay SA, DuPont, and Victrex PLC leverage vertically integrated production, proprietary polymer grades, and long-term supply agreements with original equipment manufacturers (OEMs) as key competitive differentiators. Strategic initiatives include capacity expansion in Asia Pacific, investment in bioresorbable polymer R&D, and acquisitions targeting specialty compounders. Business models are evolving from commodity supply toward value-added polymer solution partnerships, with companies offering technical co-development services, regulatory filing support, and tailored compound formulations for device manufacturers.
Key Market Developments
- March 2024: Victrex PLC announced the commercial launch of its new PEEK-based biomaterial grade specifically certified for long-term implantable spinal fusion devices, targeting the rapidly growing minimally invasive spine surgery segment.
- September 2024: Solvay SA expanded its Specialty Polymers division by inaugurating a dedicated medical-grade polymer compounding facility in Changshu, China, to serve the surging Asia Pacific medical device manufacturing demand.
- January 2025: Evonik Industries AG signed a strategic collaboration with a leading European orthopedic OEM to co-develop next-generation PEEK-Optima composite materials for load-bearing implant applications, reinforcing R&D-led market positioning.
Companies Covered in High Performance Medical Plastic Market
- Solvay SA
- SABIC
- DuPont
- Mitsubishi Chemical Corp
- Evonik Industries AG
- Saint-Gobain Performance Plastics
- Celanese Corporation
- Ensinger GmbH
- Arkema SA
- Victrex PLC
- Trelleborg AB
- Raumedic AG
- Tekni-Plex
- BASF SE
- DSM Engineering Materials (now Avantium/dsm-firmenich)
- RTP Company
- Nolato AB
Market Segmentation
By Product Type
- Fluoropolymers (FP)
- High Performance Polyamides (HPPA)
- Polyaryletherketones (PAEK)
- Polyphenylene Sulfides (PPS)
- Polyetherimide (PEI)
- Others
By Application
- Medical Equipment and Tools
- Medical Supplies
- Drug Delivery
- Prosthesis & Implants
- Therapeutic Systems
- Others
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
- Executive Summary
- Global High Performance Medical Plastic Market Snapshot
- Future Projections
- Key Market Trends
- Regional Snapshot, by Value, 2026
- Analyst Recommendations
- Market Overview
- Market Definitions and Segmentations
- Market Dynamics
- Drivers
- Restraints
- Market Opportunities
- Value Chain Analysis
- COVID-19 Impact Analysis
- Porter's Five Forces Analysis
- Impact of Russia-Ukraine Conflict
- PESTLE Analysis
- Regulatory Analysis
- Price Trend Analysis
- Current Prices and Future Projections, 2025-2033
- Price Impact Factors
- Global High Performance Medical Plastic Market Outlook, 2020 - 2033
- Global High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Fluoropolymers(FP)
- High Performance Polyamides(HPPA)
- Polyaryletherketones(PAEK)
- Polyphenylene Sulfides(PPS)
- Polyetherimide(PEI)
- Others
- Global High Performance Medical Plastic Market Outlook, by Application, Value (US$ Bn), 2020-2033
- Medical Equipments and Tools
- Medical Supplies
- Drug Delivery
- Prosthesis & Implants
- Therapeutic System
- Others
- Global High Performance Medical Plastic Market Outlook, by Region, Value (US$ Bn), 2020-2033
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
- Global High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- North America High Performance Medical Plastic Market Outlook, 2020 - 2033
- North America High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Fluoropolymers(FP)
- High Performance Polyamides(HPPA)
- Polyaryletherketones(PAEK)
- Polyphenylene Sulfides(PPS)
- Polyetherimide(PEI)
- Others
- North America High Performance Medical Plastic Market Outlook, by Application, Value (US$ Bn), 2020-2033
- Medical Equipments and Tools
- Medical Supplies
- Drug Delivery
- Prosthesis & Implants
- Therapeutic System
- Others
- North America High Performance Medical Plastic Market Outlook, by Country, Value (US$ Bn), 2020-2033
- S. High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- S. High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Canada High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Canada High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- BPS Analysis/Market Attractiveness Analysis
- North America High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Europe High Performance Medical Plastic Market Outlook, 2020 - 2033
- Europe High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Fluoropolymers(FP)
- High Performance Polyamides(HPPA)
- Polyaryletherketones(PAEK)
- Polyphenylene Sulfides(PPS)
- Polyetherimide(PEI)
- Others
- Europe High Performance Medical Plastic Market Outlook, by Application, Value (US$ Bn), 2020-2033
- Medical Equipments and Tools
- Medical Supplies
- Drug Delivery
- Prosthesis & Implants
- Therapeutic System
- Others
- Europe High Performance Medical Plastic Market Outlook, by Country, Value (US$ Bn), 2020-2033
- Germany High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Germany High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Italy High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Italy High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- France High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- France High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- K. High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- K. High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Spain High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Spain High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Russia High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Russia High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Rest of Europe High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Rest of Europe High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- BPS Analysis/Market Attractiveness Analysis
- Europe High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Asia Pacific High Performance Medical Plastic Market Outlook, 2020 - 2033
- Asia Pacific High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Fluoropolymers(FP)
- High Performance Polyamides(HPPA)
- Polyaryletherketones(PAEK)
- Polyphenylene Sulfides(PPS)
- Polyetherimide(PEI)
- Others
- Asia Pacific High Performance Medical Plastic Market Outlook, by Application, Value (US$ Bn), 2020-2033
- Medical Equipments and Tools
- Medical Supplies
- Drug Delivery
- Prosthesis & Implants
- Therapeutic System
- Others
- Asia Pacific High Performance Medical Plastic Market Outlook, by Country, Value (US$ Bn), 2020-2033
- China High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- China High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Japan High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Japan High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- South Korea High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- South Korea High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- India High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- India High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Southeast Asia High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Southeast Asia High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Rest of SAO High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Rest of SAO High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- BPS Analysis/Market Attractiveness Analysis
- Asia Pacific High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Latin America High Performance Medical Plastic Market Outlook, 2020 - 2033
- Latin America High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Fluoropolymers(FP)
- High Performance Polyamides(HPPA)
- Polyaryletherketones(PAEK)
- Polyphenylene Sulfides(PPS)
- Polyetherimide(PEI)
- Others
- Latin America High Performance Medical Plastic Market Outlook, by Application, Value (US$ Bn), 2020-2033
- Medical Equipments and Tools
- Medical Supplies
- Drug Delivery
- Prosthesis & Implants
- Therapeutic System
- Others
- Latin America High Performance Medical Plastic Market Outlook, by Country, Value (US$ Bn), 2020-2033
- Brazil High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Brazil High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Mexico High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Mexico High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Argentina High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Argentina High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Rest of LATAM High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Rest of LATAM High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- BPS Analysis/Market Attractiveness Analysis
- Latin America High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Middle East & Africa High Performance Medical Plastic Market Outlook, 2020 - 2033
- Middle East & Africa High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Fluoropolymers(FP)
- High Performance Polyamides(HPPA)
- Polyaryletherketones(PAEK)
- Polyphenylene Sulfides(PPS)
- Polyetherimide(PEI)
- Others
- Middle East & Africa High Performance Medical Plastic Market Outlook, by Application, Value (US$ Bn), 2020-2033
- Medical Equipments and Tools
- Medical Supplies
- Drug Delivery
- Prosthesis & Implants
- Therapeutic System
- Others
- Middle East & Africa High Performance Medical Plastic Market Outlook, by Country, Value (US$ Bn), 2020-2033
- GCC High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- GCC High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- South Africa High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- South Africa High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Egypt High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Egypt High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Nigeria High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Nigeria High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- Rest of Middle East High Performance Medical Plastic Market Outlook, by Product Type, 2020-2033
- Rest of Middle East High Performance Medical Plastic Market Outlook, by Application, 2020-2033
- BPS Analysis/Market Attractiveness Analysis
- Middle East & Africa High Performance Medical Plastic Market Outlook, by Product Type, Value (US$ Bn), 2020-2033
- Competitive Landscape
- Company Vs Segment Heatmap
- Company Market Share Analysis, 2025
- Competitive Dashboard
- Company Profiles
- Solvay SA
- Company Overview
- Product Portfolio
- Financial Overview
- Business Strategies and Developments
- SABIC
- DUPont
- Mitsubishi Chemical Corp
- Evonik Industries AG
- Saint-Gobain Performance Plastics
- Celanese Corporation
- Ensimger GmbH
- Arkema SA
- Victrex PLC
- Trelleborg AB
- Raumedic AG
- Tekni-Plex
- Solvay SA
- Appendix
- Research Methodology
- Report Assumptions
- Acronyms and Abbreviations
|
BASE YEAR |
HISTORICAL DATA |
FORECAST PERIOD |
UNITS |
|||
|
2025 |
2019 - 2024 |
2026 - 2033 |
Value: US$ Million |
|||
|
REPORT FEATURES |
DETAILS |
|
By Product Type Coverage |
|
|
By Application Coverage |
|
|
Geographical Coverage |
|
|
Leading Companies |
|
|
Report Highlights |
Key Market Indicators, Macro-micro economic impact analysis, Technological Roadmap, Key Trends, Driver, Restraints, and Future Opportunities & Revenue Pockets, Porter’s 5 Forces Analysis, Historical Trend (2019-2024), Market Estimates and Forecast, Market Dynamics, Industry Trends, Competition Landscape, Category, Region, Country-wise Trends & Analysis, COVID-19 Impact Analysis (Demand and Supply Chain) |
