Global Cell & Gene Therapy Bioanalytical Testing Services Market Forecast
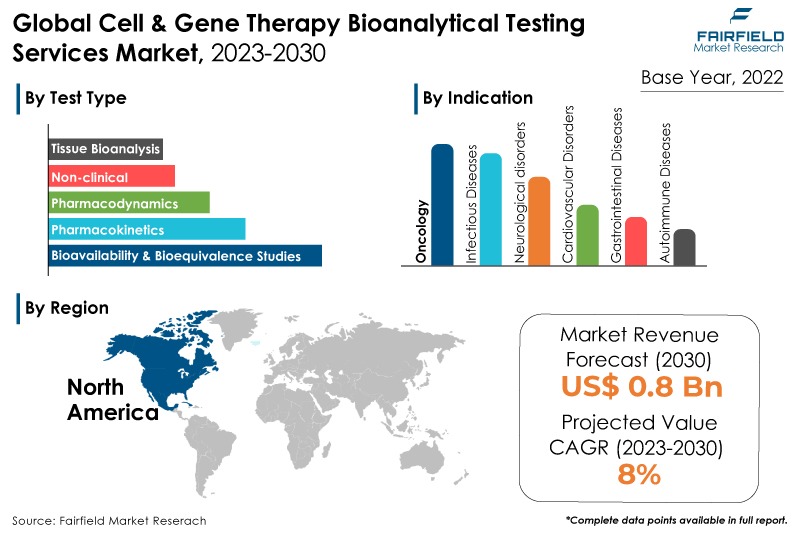
- Global market valuiation to rise high a CAGR of 8% during 2023 - 2030
- Market size likely to reach approximately US$0.8 Bn by the end of 2030
Market Analysis in Brief
Cell and gene therapy bioanalytical testing services refer to specialised laboratory services that analyse cellular and genetic components of therapeutic products. Cell and gene therapies are innovative medical treatments that involve manipulating or modifying a patient's cells or genes to treat or prevent diseases. Bioanalytical testing services ensure these therapies' safety, efficacy, and quality by conducting various tests and analyses on the therapeutic products. Cell and gene therapies are experiencing rapid advancements, with innovative treatments being developed for various diseases, including genetic disorders, cancer, and autoimmune conditions.
As more therapies reach clinical trials and approval stages, the demand for comprehensive bioanalytical testing services increases. Regulatory agencies such as the European Medicines Agency (EMA), and the US Food and Drug Administration (FDA) impose stringent requirements on cell and gene therapies' quality, safety, and efficacy. Bioanalytical testing services are crucial for meeting these regulatory standards and obtaining necessary approvals. Cell and gene therapies involve manipulating a patient's cells or genetic material. Ensuring these therapies' safety is paramount; rigorous testing is required to identify potential risks and adverse effects.
Key Report Findings
- The market for cell & gene therapy bioanalytical testing services will demonstrate massive expansion in revenue between 2023 and 2030.
- Demand for bioavailability & bioequivalence studies remains higher in the cell & gene therapy bioanalytical testing services market.
- Demand for non-clinical products remains higher in the cell & gene therapy bioanalytical testing services market.
- Cell therapy held the highest market share in 2022.
- North America will continue to lead, whereas the Asia Pacific market will experience the strongest growth till 2030.
Growth Drivers
Rapid Advancements in Cell & Gene Therapies
Rapid advancements in cell and gene therapies significantly fuel the growth of the cell & gene therapy bioanalytical testing services market. These advancements create a higher demand for rigorous and comprehensive testing to ensure these innovative therapies' safety, efficacy, and quality.
As cell and gene therapies become more sophisticated, involving intricate genetic modifications and cellular manipulations, the need for specialised testing services increases. Comprehensive bioanalytical testing is essential to assess complex interactions and outcomes of these therapies.
Advances in cell and gene therapies lead to the development of new treatment approaches for previously untreatable or poorly managed diseases. Before reaching patients, bioanalytical testing ensures these novel therapies meet the highest safety and efficacy standards. The rapid rate of cell and gene therapy research translates into more therapies entering clinical trials.
Bioanalytical testing services are crucial in monitoring these trials, assessing patient responses, and generating valuable data for regulatory submissions. Regulatory agencies are working to expedite the approval process for promising cell and gene therapies.
However, this acceleration requires thorough and reliable testing to demonstrate the therapies' safety and efficacy within shorter timeframes. Many cell and gene therapies are personalised and tailored to individual patients. Bioanalytical testing ensures accurate characterisation of patient-specific materials and validates the therapy's identity and quality.
The rapid progress in cell and gene therapies drives the demand for thorough bioanalytical testing services. These services are essential to support the development, regulatory approval, clinical trials, and post-market monitoring of these innovative therapies, ensuring they meet the highest safety and efficacy standards.
Regulatory Requirements
Regulatory requirements drive the cell & gene therapy bioanalytical testing services market. The stringent standards and guidelines set by regulatory agencies ensure cell and gene therapies' safety, efficacy, and quality before they are approved for clinical use. As a result, the demand for comprehensive and specialised bioanalytical testing services increases.
Regulatory agencies prioritise patient safety, such as the US Food and Drug Administration (FDA), and the European Medicines Agency (EMA). They require rigorous testing to identify potential risks, adverse effects, and toxicity associated with cell and gene therapies. Regulatory bodies demand data that demonstrate the efficacy of cell and gene therapies. Bioanalytical testing provides evidence of the therapy's intended effects and contributes to patient benefit-risk assessment.
Regulatory agencies mandate stringent quality control measures to ensure consistent and reliable therapeutic products. Bioanalytical testing services validate the therapies' quality, identity, and purity, meeting regulatory criteria. Any changes to the manufacturing process or therapy components require extensive testing to ensure the therapy's consistency and effectiveness.
Regulatory agencies mandate comparability studies, which drive the need for bioanalytical testing. Cell and gene therapies must undergo rigorous clinical trials to obtain regulatory approval. Bioanalytical testing generates critical data used to evaluate the safety and efficacy of these therapies during trials.
Regulatory agencies require comprehensive characterisation of cell and gene therapies, including the specific cell types, genetic modifications, and vector components. Bioanalytical testing verifies product identity and composition. Cell and gene therapies need to demonstrate stability over their intended shelf life. Regulatory agencies require data on the therapy's stability profile, obtained through thorough bioanalytical testing.
Growth Challenges
Lack of Standardisation in Testing Protocols
Like any other industry, the market of cell & gene therapy bioanalytical testing services faces various challenges and restraints that can impact its growth and development. Cell and gene therapies are often complex and involve intricate cellular and genetic modifications. This complexity poses challenges for developing accurate and standardised testing methodologies.
The lack of standardised testing protocols across several therapies and manufacturers can lead to consistency in testing results. Harmonising testing methods can be challenging due to the unique nature of each therapy. The specialised nature of bioanalytical testing for cell and gene therapies requires skilled professionals with expertise in diverse scientific fields. More qualified experts are needed to ensure the availability of testing services.
Regulatory agencies require rigorous testing to ensure cell and gene therapies' safety, efficacy, and quality. Meeting these regulatory standards can be time-consuming and resource-intensive for testing service providers.
Developing and conducting bioanalytical testing services can be costly due to the advanced equipment, skilled personnel, and specialised facilities required. Balancing competitive pricing while maintaining quality can be a challenge.
Overview of Key Segments
Bioavailability and Bioequivalence Studies Prominent
Bioavailability and bioequivalence studies play an important role in the cell & gene therapy bioanalytical testing services market by providing valuable insights into cell and gene therapies' pharmacokinetics, safety, and effectiveness. These studies offer several benefits contributing to these therapies' development, regulatory approval, and successful clinical implementation.
Bioavailability studies help determine how much of the administered therapy reaches the bloodstream and is available for the body. This information is crucial for understanding the therapy's pharmacokinetics, including absorption, distribution, metabolism, and excretion. Bioavailability studies provide data on the therapy's rate and extent of absorption. This information helps optimise dosing strategies, ensuring that the therapy reaches therapeutic levels in the body for the desired duration.
Bioequivalence studies compare the bioavailability of a generic or biosimilar therapy to the original reference therapy. These studies ensure that generic or biosimilar versions regarding safety and efficacy are equivalent. Regulatory agencies require bioavailability and bioequivalence data to demonstrate the safety and efficacy of therapies, especially for generic versions. These studies are essential for gaining regulatory approvals for commercialisation.
Bioavailability studies contribute to quality control by ensuring consistent delivery of the therapy. Variations in bioavailability can impact the therapy's performance and patient outcomes. Bioavailability and bioequivalence studies provide critical insights into how cell and gene therapies behave in the body, ensuring their safety, efficacy, and consistent performance. These studies are essential for regulatory compliance, treatment optimisation, and successful clinical implementation of cell and gene therapies.
Demand for Testing Services in Demand
Oncology-focused bioanalytical testing services offer several cancer treatment and research benefits in the Cell & Gene Therapy market. These services contribute to developing, evaluating, and optimising cell and gene therapies targeting various forms of cancer. Oncology-focused bioanalytical testing helps develop cell and gene therapies targeting cancer cells, minimising damage to healthy tissues and reducing side effects.
Bioanalytical testing allows for the customisation of therapies based on the patient's unique genetic and molecular profile, improving treatment efficacy and patient outcomes. These studies provide insights into how cancer patients absorb, distribute, metabolise, and excrete the therapy. This information helps optimise dosing regimens for maximum therapeutic benefit.
Bioanalytical testing allows for monitoring the concentration of the therapeutic agent in a patient's blood over time, ensuring that the therapy remains within the therapeutic range. In immunotherapies, bioanalytical testing helps assess the potential for immune responses to the therapy, which is crucial for treatment safety and efficacy. Bioanalytical testing helps measure treatment response by analysing biomarkers that indicate tumor progression or regression, guiding treatment adjustments.
In the field of oncology, where the need for effective and targeted therapies is critical, bioanalytical testing services play an instrumental role in advancing the development and optimisation of cell and gene therapies. These services enable researchers and clinicians to tailor treatments to individual patients, monitor responses, and refine therapeutic approaches for improved outcomes.
Growth Opportunities Across Regions
North America at the Forefront
North America is a leader in the cell & gene therapy bioanalytical testing services market. Several factors have contributed to North America's prominent position in this sector. North America, particularly the United States, has a well-established and vibrant biotechnology industry.
The region is home to many biotech companies actively involved in developing and commercialising cell and gene therapies. Leading research institutions, universities, and medical centers in North America have been at the forefront of cell and gene therapy research. This research excellence has driven the demand for bioanalytical testing services to support preclinical and clinical studies.
North America has been an early adopter of novel medical therapies, including cell and gene therapies. The region's regulatory agencies have been relatively receptive to these therapies, creating an environment conducive to their development and testing.
Many global clinical trials for cell and gene therapies are conducted in North America. This high level of clinical trial activity requires extensive bioanalytical testing to assess safety, efficacy, and regulatory compliance.
Regulatory agencies like the US Food and Drug Administration (FDA) have proactively created regulatory pathways for cell and gene therapies. Their guidance and expertise have facilitated the development and testing of these therapies. The success and innovation generated in North America's biotechnology sector have a global impact, influencing developments and collaborations in other regions.
Asia Pacific Represents a Highly Profitable Pocket
Due to several key factors, Asia Pacific has emerged as a growing and potentially lucrative market for cell & gene therapy bioanalytical testing services. Asia Pacific is experiencing significant economic growth, leading to increased investment in healthcare infrastructure, research, and development. This growth provides a fertile ground for developing and adopting innovative therapies like cell and gene therapies.
Many countries in the Asia Pacific have been investing in biotechnology research and development. The region has seen the emergence of biotech companies focused on developing and commercialising advanced therapies. The region has attracted substantial investment from local and global investors, fostering the growth of the cell and gene therapy sector. This funding supports research, clinical trials, and testing services.
Asia Pacific's diverse population and patient pool make it an attractive location for conducting clinical trials. As cell and gene therapies improve through clinical phases, the demand for comprehensive bioanalytical testing services increases. Some countries in the Asia Pacific have been working to establish clear regulatory frameworks for cell and gene therapies. Regulatory advancements create a more predictable environment for testing services and commercialisation.
Cell & Gene Therapy Bioanalytical Testing Services Market: Competitive Landscape
Some of the leading players at the forefront in the cell & gene therapy bioanalytical Testing Services market space include BioAgilytix Labs, KCAS Bioanalytical Services, Laboratory Corporation of America Holdings, Pharmaceutical Product Development, Inc., Prolytix, Pharmaron, Charles River Laboratories, Syneos Health, SGS SA, and CD BioSciences.
Recent Notable Developments
In February 2023, KCAS Bioanalytical and Biomarker Services, a Vitruvian Partners portfolio Company, announced the acquisition of FlowMetric, LLC. The acquisition of FlowMetric LLC by KCAS empowers KCAS to address customer demands better and expedite its swift expansion, solidifying its position as the preferred clinical research organisation (CRO) for outsourced cell and gene therapy initiatives.
In March 2021, Charles River successfully finalised the acquisition of Cognate BioServices, a move to establish a top-tier scientific collaborator in cell and gene therapy advancement. Cognate BioServices is a leading contract development and manufacturing organisation (CDMO) specialising in cell and gene therapies. Their comprehensive manufacturing solutions encompass the production of cell therapies, plasmid DNA, and other critical components within the CDMO value chain.
Global Cell & Gene Therapy Bioanalytical Testing Services Market is Segmented as Below:
By Test Type
- Bioavailability Bioavailability & Bioequivalence Studies
- Pharmacokinetics
- Pharmacodynamics
- Non-clinical
- Tissue Bioanalysis
- Miscellaneous
By Therapy Type
- Gene Therapy
- Gene-Modified Cell Therapy
- Cell Therapy
By End user
- Non-Clinical Contract Manufacturing Organizations (CMO)
- Pharma and Biopharma Companies
- Contract Research Organizations (CRO)
By Indication
- Oncology Cancer
- Infectious Diseases
- Neurological disorders
- Cardiovascular Disorders
- Gastrointestinal Diseases
- Autoimmune Diseases
- Others
By Geographic Coverage
- North America
- The U.S.
- Canada
- Europe
- Germany
- The U.K.
- France
- Italy
- Turkey
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Southeast Asia
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Argentina
- Rest of Latin America
- Middle East & Africa
- GCC
- South Africa
- Egypt
- Nigeria
- Rest of the Middle East & Africa
1. Executive Summary
1.1. Global Cell & Gene Therapy Bioanalytical Testing Services Market Snapshot
1.2. Future Projections
1.3. Key Market Trends
1.4. Regional Snapshot, by Value, 2022
1.5. Analyst Recommendations
2. Market Overview
2.1. Market Definitions and Segmentations
2.2. Market Dynamics
2.2.1. Drivers
2.2.2. Restraints
2.2.3. Market Opportunities
2.3. Value Chain Analysis
2.4. Porter’s Five Forces Analysis
2.5. COVID-19 Impact Analysis
2.5.1. Supply
2.5.2. Demand
2.6. Impact of Ukraine-Russia Conflict
2.7. Economic Overview
2.7.1. World Economic Projections
2.8. PESTLE Analysis
3. Global Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, 2018 - 2030
3.1. Global Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Test Type, Value (US$ Mn), 2018 - 2030
3.1.1. Key Highlights
3.1.1.1. Bioavailability & Bioequivalence Studies
3.1.1.2. Pharmacokinetics
3.1.1.3. Pharmacodynamics
3.1.1.4. Non-clinical
3.1.1.5. Tissue Bioanalysis
3.1.1.6. Misc.
3.2. Global Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Therapy Type, Value (US$ Mn), 2018 - 2030
3.2.1. Key Highlights
3.2.1.1. Gene Therapy
3.2.1.2. Gene-Modified Cell Therapy
3.2.1.3. Cell Therapy
3.3. Global Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by End user, Value (US$ Mn), 2018 - 2030
3.3.1. Key Highlights
3.3.1.1. Contract Manufacturing Organizations (CMO)
3.3.1.2. Pharma and Biopharma Companies
3.3.1.3. Contract Research Organizations (CRO)
3.4. Global Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Indication, Value (US$ Mn), 2018 - 2030
3.4.1. Key Highlights
3.4.1.1. Cancer
3.4.1.2. Infectious Diseases
3.4.1.3. Neurological disorders
3.4.1.4. Cardiovascular Disorders
3.4.1.5. Gastrointestinal Diseases
3.4.1.6. Autoimmune Diseases
3.4.1.7. Others
3.5. Global Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Region, Value (US$ Mn), 2018 - 2030
3.5.1. Key Highlights
3.5.1.1. North America
3.5.1.2. Europe
3.5.1.3. Asia Pacific
3.5.1.4. Latin America
3.5.1.5. Middle East & Africa
4. North America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, 2018 - 2030
4.1. North America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Test Type, Value (US$ Mn), 2018 - 2030
4.1.1. Key Highlights
4.1.1.1. Bioavailability & Bioequivalence Studies
4.1.1.2. Pharmacokinetics
4.1.1.3. Pharmacodynamics
4.1.1.4. Non-clinical
4.1.1.5. Tissue Bioanalysis
4.1.1.6. Misc.
4.2. North America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Therapy Type, Value (US$ Mn), 2018 - 2030
4.2.1. Key Highlights
4.2.1.1. Gene Therapy
4.2.1.2. Gene-Modified Cell Therapy
4.2.1.3. Cell Therapy
4.3. North America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by End user, Value (US$ Mn), 2018 - 2030
4.3.1. Key Highlights
4.3.1.1. Contract Manufacturing Organizations (CMO)
4.3.1.2. Pharma and Biopharma Companies
4.3.1.3. Contract Research Organizations (CMO)
4.4. North America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Indication, Value (US$ Mn), 2018 - 2030
4.4.1. Key Highlights
4.4.1.1. Cancer
4.4.1.2. Infectious Diseases
4.4.1.3. Neurological disorders
4.4.1.4. Cardiovascular Disorders
4.4.1.5. Gastrointestinal Diseases
4.4.1.6. Autoimmune Diseases
4.4.1.7. Others
4.4.2. Market Attractiveness Analysis
4.5. North America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Country, Value (US$ Mn), 2018 - 2030
4.5.1. Key Highlights
4.5.1.1. U.S. Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
4.5.1.2. U.S. Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
4.5.1.3. U.S. Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
4.5.1.4. U.S. Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
4.5.1.5. Canada Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
4.5.1.6. Canada Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
4.5.1.7. Canada Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
4.5.1.8. Canada Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
4.5.2. BPS Analysis/Market Attractiveness Analysis
5. Europe Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, 2018 - 2030
5.1. Europe Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Test Type, Value (US$ Mn), 2018 - 2030
5.1.1. Key Highlights
5.1.1.1. Bioavailability & Bioequivalence Studies
5.1.1.2. Pharmacokinetics
5.1.1.3. Pharmacodynamics
5.1.1.4. Non-clinical
5.1.1.5. Tissue Bioanalysis
5.1.1.6. Misc.
5.2. Europe Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Therapy Type, Value (US$ Mn), 2018 - 2030
5.2.1. Key Highlights
5.2.1.1. Gene Therapy
5.2.1.2. Gene-Modified Cell Therapy
5.2.1.3. Cell Therapy
5.3. Europe Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by End user, Value (US$ Mn), 2018 - 2030
5.3.1. Key Highlights
5.3.1.1. Contract Manufacturing Organizations (CMO)
5.3.1.2. Pharma and Biopharma Companies
5.3.1.3. Contract Research Organizations (CMO)
5.4. Europe Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Indication, Value (US$ Mn), 2018 - 2030
5.4.1. Key Highlights
5.4.1.1. Cancer
5.4.1.2. Infectious Diseases
5.4.1.3. Neurological disorders
5.4.1.4. Cardiovascular Disorders
5.4.1.5. Gastrointestinal Diseases
5.4.1.6. Autoimmune Diseases
5.4.1.7. Others
5.4.2. Market Attractiveness Analysis
5.5. Europe Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Country, Value (US$ Mn), 2018 - 2030
5.5.1. Key Highlights
5.5.1.1. Germany Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
5.5.1.2. Germany Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
5.5.1.3. Germany Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
5.5.1.4. Germany Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
5.5.1.5. U.K. Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
5.5.1.6. U.K. Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
5.5.1.7. U.K. Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
5.5.1.8. U.K. Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
5.5.1.9. France Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
5.5.1.10. France Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
5.5.1.11. France Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
5.5.1.12. France Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
5.5.1.13. Italy Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
5.5.1.14. Italy Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
5.5.1.15. Italy Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
5.5.1.16. Italy Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
5.5.1.17. Turkey Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
5.5.1.18. Turkey Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
5.5.1.19. Turkey Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
5.5.1.20. Turkey Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
5.5.1.21. Russia Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
5.5.1.22. Russia Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
5.5.1.23. Russia Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
5.5.1.24. Russia Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
5.5.1.25. Rest of Europe Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
5.5.1.26. Rest of Europe Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
5.5.1.27. Rest of Europe Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
5.5.1.28. Rest of Europe Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
5.5.2. BPS Analysis/Market Attractiveness Analysis
6. Asia Pacific Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, 2018 - 2030
6.1. Asia Pacific Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Test Type, Value (US$ Mn), 2018 - 2030
6.1.1. Key Highlights
6.1.1.1. Bioavailability & Bioequivalence Studies
6.1.1.2. Pharmacokinetics
6.1.1.3. Pharmacodynamics
6.1.1.4. Non-clinical
6.1.1.5. Tissue Bioanalysis
6.1.1.6. Misc.
6.2. Asia Pacific Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Therapy Type, Value (US$ Mn), 2018 - 2030
6.2.1. Key Highlights
6.2.1.1. Gene Therapy
6.2.1.2. Gene-Modified Cell Therapy
6.2.1.3. Cell Therapy
6.3. Asia Pacific Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by End user, Value (US$ Mn), 2018 - 2030
6.3.1. Key Highlights
6.3.1.1. Contract Manufacturing Organizations (CMO)
6.3.1.2. Pharma and Biopharma Companies
6.3.1.3. Contract Research Organizations (CRO)
6.4. Asia Pacific Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Indication, Value (US$ Mn), 2018 - 2030
6.4.1. Key Highlights
6.4.1.1. Cancer
6.4.1.2. Infectious Diseases
6.4.1.3. Neurological disorders
6.4.1.4. Cardiovascular Disorders
6.4.1.5. Gastrointestinal Diseases
6.4.1.6. Autoimmune Diseases
6.4.1.7. Others
6.4.2. Market Attractiveness Analysis
6.5. Asia Pacific Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Country, Value (US$ Mn), 2018 - 2030
6.5.1. Key Highlights
6.5.1.1. China Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
6.5.1.2. China Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
6.5.1.3. China Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
6.5.1.4. China Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
6.5.1.5. Japan Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
6.5.1.6. Japan Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
6.5.1.7. Japan Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
6.5.1.8. Japan Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
6.5.1.9. South Korea Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
6.5.1.10. South Korea Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
6.5.1.11. South Korea Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
6.5.1.12. South Korea Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
6.5.1.13. India Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
6.5.1.14. India Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
6.5.1.15. India Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
6.5.1.16. India Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
6.5.1.17. Southeast Asia Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
6.5.1.18. Southeast Asia Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
6.5.1.19. Southeast Asia Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
6.5.1.20. Southeast Asia Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
6.5.1.21. Rest of Asia Pacific Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
6.5.1.22. Rest of Asia Pacific Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
6.5.1.23. Rest of Asia Pacific Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
6.5.1.24. Rest of Asia Pacific Cell & Gene Therapy Bioanalytical Testing Services Market by Indication, Value (US$ Mn), 2018 - 2030
6.5.2. BPS Analysis/Market Attractiveness Analysis
7. Latin America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, 2018 - 2030
7.1. Latin America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Test Type, Value (US$ Mn), 2018 - 2030
7.1.1. Key Highlights
7.1.1.1. Bioavailability & Bioequivalence Studies
7.1.1.2. Pharmacokinetics
7.1.1.3. Pharmacodynamics
7.1.1.4. Non-clinical
7.1.1.5. Tissue Bioanalysis
7.1.1.6. Misc.
7.2. Latin America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Therapy Type, Value (US$ Mn), 2018 - 2030
7.2.1. Key Highlights
7.2.1.1. Gene Therapy
7.2.1.2. Gene-Modified Cell Therapy
7.2.1.3. Cell Therapy
7.3. Latin America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by End user, Value (US$ Mn), 2018 - 2030
7.3.1. Key Highlights
7.3.1.1. Contract Manufacturing Organizations (CMO)
7.3.1.2. Pharma and Biopharma Companies
7.3.1.3. Contract Manufacturing Organizations (CMO)
7.4. Latin America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Indication, Value (US$ Mn), 2018 - 2030
7.4.1. Key Highlights
7.4.1.1. Cancer
7.4.1.2. Infectious Diseases
7.4.1.3. Neurological disorders
7.4.1.4. Cardiovascular Disorders
7.4.1.5. Gastrointestinal Diseases
7.4.1.6. Autoimmune Diseases
7.4.1.7. Others
7.4.2. Market Attractiveness Analysis
7.5. Latin America Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Country, Value (US$ Mn), 2018 - 2030
7.5.1. Key Highlights
7.5.1.1. Brazil Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
7.5.1.2. Brazil Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
7.5.1.3. Brazil Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
7.5.1.4. Brazil Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
7.5.1.5. Mexico Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
7.5.1.6. Mexico Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
7.5.1.7. Mexico Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
7.5.1.8. Mexico Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
7.5.1.9. Argentina Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
7.5.1.10. Argentina Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
7.5.1.11. Argentina Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
7.5.1.12. Argentina Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
7.5.1.13. Rest of Latin America Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
7.5.1.14. Rest of Latin America Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
7.5.1.15. Rest of Latin America Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
7.5.1.16. Rest of Latin America Cell & Gene Therapy Bioanalytical Testing Services Market by Indication, Value (US$ Mn), 2018 - 2030
7.5.2. BPS Analysis/Market Attractiveness Analysis
8. Middle East & Africa Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, 2018 - 2030
8.1. Middle East & Africa Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Test Type, Value (US$ Mn), 2018 - 2030
8.1.1. Key Highlights
8.1.1.1. Bioavailability & Bioequivalence Studies
8.1.1.2. Pharmacokinetics
8.1.1.3. Pharmacodynamics
8.1.1.4. Non-clinical
8.1.1.5. Tissue Bioanalysis
8.1.1.6. Misc.
8.2. Middle East & Africa Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Therapy Type, Value (US$ Mn), 2018 - 2030
8.2.1. Key Highlights
8.2.1.1. Gene Therapy
8.2.1.2. Gene-Modified Cell Therapy
8.2.1.3. Cell Therapy
8.3. Middle East & Africa Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by End user, Value (US$ Mn), 2018 - 2030
8.3.1. Key Highlights
8.3.1.1. Contract Manufacturing Organizations (CMO)
8.3.1.2. Pharma and Biopharma Companies
8.3.1.3. Contract Research Organizations (CMO)
8.4. Middle East & Africa Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Indication, Value (US$ Mn), 2018 - 2030
8.4.1. Key Highlights
8.4.1.1. Cancer
8.4.1.2. Infectious Diseases
8.4.1.3. Neurological disorders
8.4.1.4. Cardiovascular Disorders
8.4.1.5. Gastrointestinal Diseases
8.4.1.6. Autoimmune Diseases
8.4.1.7. Others
8.4.2. Market Attractiveness Analysis
8.5. Middle East & Africa Cell & Gene Therapy Bioanalytical Testing Services Market Outlook, by Country, Value (US$ Mn), 2018 - 2030
8.5.1. Key Highlights
8.5.1.1. GCC Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
8.5.1.2. GCC Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
8.5.1.3. GCC Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
8.5.1.4. GCC Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
8.5.1.5. South Africa Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
8.5.1.6. South Africa Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
8.5.1.7. South Africa Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
8.5.1.8. South Africa Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
8.5.1.9. Egypt Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
8.5.1.10. Egypt Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
8.5.1.11. Egypt Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
8.5.1.12. Egypt Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
8.5.1.13. Nigeria Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
8.5.1.14. Nigeria Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
8.5.1.15. Nigeria Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
8.5.1.16. Nigeria Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
8.5.1.17. Rest of Middle East & Africa Cell & Gene Therapy Bioanalytical Testing Services Market by Test Type, Value (US$ Mn), 2018 - 2030
8.5.1.18. Rest of Middle East & Africa Cell & Gene Therapy Bioanalytical Testing Services Market Therapy Type, Value (US$ Mn), 2018 - 2030
8.5.1.19. Rest of Middle East & Africa Cell & Gene Therapy Bioanalytical Testing Services Market by End user, Value (US$ Mn), 2018 - 2030
8.5.1.20. Rest of Middle East & Africa Cell & Gene Therapy Bioanalytical Testing Services Market Indication, Value (US$ Mn), 2018 - 2030
8.5.2. BPS Analysis/Market Attractiveness Analysis
9. Competitive Landscape
9.1. Service Providers Vs Test Type Heatmap
9.2. Company Market Share Analysis, 2022
9.3. Competitive Dashboard
9.4. Company Profiles
9.4.1. BioAgilytix Labs
9.4.1.1. Company Overview
9.4.1.2. Product Portfolio
9.4.1.3. Financial Overview
9.4.1.4. Business Strategies and Development
9.4.2. KCAS Bioanalytical Services
9.4.2.1. Company Overview
9.4.2.2. Product Portfolio
9.4.2.3. Financial Overview
9.4.2.4. Business Strategies and Development
9.4.3. CD BioSciences
9.4.3.1. Company Overview
9.4.3.2. Product Portfolio
9.4.3.3. Financial Overview
9.4.3.4. Business Strategies and Development
9.4.4. Laboratory Corporation of America Holdings
9.4.4.1. Company Overview
9.4.4.2. Product Portfolio
9.4.4.3. Financial Overview
9.4.4.4. Business Strategies and Development
9.4.5. Pharmaceutical Product Development, Inc.
9.4.5.1. Company Overview
9.4.5.2. Product Portfolio
9.4.5.3. Financial Overview
9.4.5.4. Business Strategies and Development
9.4.6. Prolytix
9.4.6.1. Company Overview
9.4.6.2. Product Portfolio
9.4.6.3. Financial Overview
9.4.6.4. Business Strategies and Development
9.4.7. Pharmaron
9.4.7.1. Company Overview
9.4.7.2. Product Portfolio
9.4.7.3. Financial Overview
9.4.7.4. Business Strategies and Development
9.4.8. Charles River Laboratories
9.4.8.1. Company Overview
9.4.8.2. Product Portfolio
9.4.8.3. Business Strategies and Development
9.4.9. Syneos Health
9.4.9.1. Company Overview
9.4.9.2. Product Portfolio
9.4.9.3. Financial Overview
9.4.9.4. Business Strategies and Development
9.4.10. SGS S.A.
9.4.10.1. Company Overview
9.4.10.2. Product Portfolio
9.4.10.3. Financial Overview
9.4.10.4. Business Strategies and Development
10. Appendix
10.1. Research Methodology
10.2. Report Assumptions
10.3. Acronyms and Abbreviations
|
BASE YEAR |
HISTORICAL DATA |
FORECAST PERIOD |
UNITS |
|||
|
2022 |
|
2018 - 2022 |
2023 - 2030 |
Value: US$ Million |
||
|
REPORT FEATURES |
DETAILS |
|
Test Type Coverage |
|
|
Therapy Type Coverage |
|
|
End User Coverage |
|
|
Indication Coverage |
|
|
Geographical Coverage |
|
|
Leading Companies |
|
|
Report Highlights |
Key Market Indicators, Macro-micro economic impact analysis, Technological Roadmap, Key Trends, Driver, Restraints, and Future Opportunities & Revenue Pockets, Porter’s 5 Forces Analysis, Historical Trend (2019-2021), Market Estimates and Forecast, Market Dynamics, Industry Trends, Competition Landscape, Category, Region, Country-wise Trends & Analysis, COVID-19 Impact Analysis (Demand and Supply Chain) |
