Global Clinical Trials Market Forecast
- Clinical trials market size to reach approximately USD 126.2 Bn in 2033, up from USD 72.7 Bn registered in 2026
- Market valuation poised to witness a CAGR of 8% during 2026 - 2033
Quick Report Digest
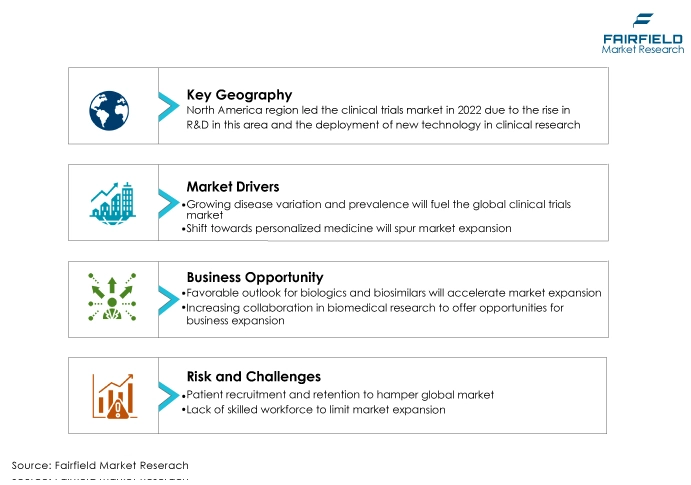
- The increasing prevalence of chronic diseases, demanding the development of novel drugs and therapies to improve the state of a patient with a chronic illness, is a significant growth driver for the clinical trials market.
- It is also anticipated that the rising R&D expenditures by major market players will have a substantial impact on the industry's expansion.
- In 2025, the phase III category dominated the industry. The main factor contributing to phase 3's significant market share is its widespread implementation. Over the estimated time, additional variables such as high costs and the growing tendency of outsourcing are anticipated to increase demand.
- In terms of market share for clinical trials globally, the interventional segment is anticipated to dominate. The significant revenue share of interventional studies is mostly due to the rising prevalence of new viral illnesses and ongoing technological advancements in clinical trials.
- In 2025, the oncology category controlled the market. The main causes of oncology's large share include ongoing research into cancer treatments and the rising need for precision medicine.
- The clinical trial data management services category is highly prevalent in the market for clinical trials. The primary drivers influencing sector growth include the increasing number of clinical trials, technical advancements in clinical trial monitoring, and strict regulations pertaining to data management & security.
- The North American region is anticipated to account for the largest share of the global clinical trials market. The rise in R&D in this area and the deployment of new technology in clinical research are both responsible for this.
- The market for clinical trials is expanding in Asia Pacific. A growing number of biotechnology businesses selected the Asia Pacific region for COVID-19 investigations because of the region's sizable patient pool and efficient procedures.
A Look Back and a Look Forward - Comparative Analysis
The ongoing efforts made by pharmaceutical, biotechnology, and medical device businesses for breakthroughs are what primarily fuel the clinical trial market. The globalisation of clinical trials, the quick development of related technologies, and the rising demand for CROs to perform clinical trials are further factors influencing the market's growth.
The market witnessed staggered growth during the historical period 2018 – 2022. The healthcare sector was under tremendous strain as a result of the growing demand for new medications to treat chronic illnesses like cancer, respiratory problems, diabetes, cardiovascular diseases, and others.
The market was being driven by the COVID-19 pandemic and the rising demand for creating an effective therapy. The measures undertaken by the government in various regions have also helped the market expand.
To satisfy the rising need for the treatment of chronic diseases, registration of trials has been expanding at a notable rate each year. The rise of the clinical trials business will be anchored by the dramatically rising number of projects launched by numerous government and non-government organisations. The increasing funding for numerous clinical trials from government funding organisations and pharmaceutical corporations will also impact the dynamics of the sector.
Key Growth Determinants
- Growing Disease Variation and Prevalence
The clinical trial market is predicted to continue to grow as disease incidence and prevalence continue to rise. The illness profiles of the global population are diverse, with rising nations having the most diversified disease profiles. This is anticipated to help clinical trials for new or rare diseases that would not have otherwise secured sponsors.
If there were more patients with a certain issue, biopharmaceutical companies would be encouraged to increase their participation in clinical trials for that disease area. A diverse population would also make patient recruiting simpler and the clinical trial process go more quickly.
In the US, uncommon diseases are referred to as "Orphan diseases," and biopharmaceutical companies that sponsor clinical trials for Orphan drugs are compensated for their efforts. This trend is most likely to benefit clinical trials for uncommon diseases, growing the market for clinical trials worldwide.
- Rapid Shift Toward Personalised Medicine
In the clinical trial market, a positive paradigm shift toward personalised treatment is anticipated. The traditional clinical trial procedure involves thousands of participants, whereas personalised medicine will concentrate solely on the impact of medications on specific patients over a predetermined length of time.
Due to the conventional clinical trial methodology, very few drugs that are still in the development phase of testing complete all rounds. Drugs that are now in development but will never be used because of the "one size fits all" approach are being prevented from entering the market.
The number of medications passing all stages of the clinical trial procedure is anticipated to rise with the application of pharmacogenetics in the process. It is anticipated that the move towards personalised medicine would enhance the use of pharmacogenetics throughout the clinical trial phase, hence expanding the available medication pool.
The biopharmaceutical industry is anticipated to increase its investment in the clinical trial stage as a result of this trend.
- Growing Drug Pipelines, and Investments in Pharmaceutical R&D
Companies in the pharmaceutical, biopharmaceutical, and medical device sectors invest heavily in the creation of cutting-edge medicines and technologies. With a focus on creating both high-quality and unique products, this industry invests heavily in R&D.
Leading pharmaceutical businesses are improving R&D efficiency, according to recent trends, by making significant R&D investments for long-term returns and encouraging joint research efforts.
A shift towards comprehensive integrated or functionally outsourced services covering the entire process of drug discovery and development, from early-stage development to late-stage development phase, is being driven by the rising R&D investments within the pharmaceutical and biopharmaceutical sectors. A sizable number of pharmaceutical and biopharmaceutical businesses currently outsource their testing functions during R&D to CROs in order to increase profitability, adhere to strict drug development timeframes, and achieve cost reductions.
Recent collaborations between well-known pharmaceutical companies, and CROs make this tendency particularly clear. Throughout the forecast period, these trends are anticipated to dominate the market.
Major Growth Barriers
- Patient Recruitment and Retention
Finding the correct patient population is challenging because attention has turned to hereditary and rare illnesses. Additionally, the lack of patient understanding of clinical trials makes it challenging for CROs to operate. Improper patient recruitment may influence the scientific and economic viability.
Patients' concerns about side effects, illiteracy, language barriers for geographically specialised clinical trials, and the lack of documentation of the consent procedure are other reasons that contribute to poor patient recruitment.
- Lack of Skilled Workforce
Clinical research organisations (CROs) have difficulty attracting and retaining qualified personnel. For competent and experienced scientists, they must compete with pharmaceutical businesses, biotech companies, manufacturers of medical equipment, and academic research institutions. These businesses must provide greater salaries and benefits in order to compete effectively.
However, this may have an impact on their earnings and performance, particularly for small-scale clinical trial service providers. The adoption of new technologies and processes may be hampered by this talent gap, which could also limit industry expansion in the years to come.
Key Trends and Opportunities to Look at
- Augmenting R&D Investments by Pharma and Biotech Companies
The development of novel technology and medicines continues to get significant funding from numerous medical device, pharmaceutical, and biopharmaceutical businesses. Particularly in the pharmaceutical industry, significant investments have been made in R&D projects for the creation of innovative treatments.
Pharmaceutical and biotechnology businesses are putting more of an emphasis on increasing their R&D productivity by funding R&D. In order to execute these trials quickly and smoothly, many companies have started outsourcing their R&D activities to CRO firms.
- Favourable Outlook for Biologics and Biosimilars
In recent years, the fields of pharmaceuticals and biotechnology have expanded quickly. R&D initiatives focused on developing novel pharmaceuticals and biological substances for treating a variety of ailments have received notable expenditures. Numerous novel medications and biologics are undergoing clinical trials right now.
The need for clinical trial services is anticipated to increase significantly due to the ongoing influx of novel pharmaceutical items in the pipeline. The COVID-19 effect resulted in a general decline in US FDA approvals in 2022, but the biologics industry was untouched, recording the approval of 15 novel compounds and retaining momentum from past years.
- Increasing Collaborations in Biomedical Research
The market for clinical trials worldwide is anticipated to be further boosted by the trend towards combination studies and collaborations in clinical trials. Biopharmaceutical businesses are now forging alliances with one another to pool resources and spread the risk of high costs as a result of the high cost of medication development.
How Does the Regulatory Scenario Shape this Industry?
The universal adoption of best practices raises clinical data credibility and makes it acceptable to regulatory bodies all around the world. The European Union, Japan, the United States, Australia, Canada, the Nordic nations, and the World Health Organisation provide a uniform standard under International Conference of Harmonisation of Technical Requirements for Registration of Medicines and Devices for Human Use.
The ICH-Efficacy recommendations concern clinical trials involving human subjects. The Declaration of Helsinki (DoH), a statement of ethical principles, was created by the World Medical Association (WMA) to serve as a guide for doctors and other individuals involved in medical research involving human people.
Furthermore, in the U.S., U.S. pharmaceutical and medical device businesses perform clinical trials as part of an application to the FDA for permission to market a medicine or device. Unless the product is exempt from this requirement, medical device businesses looking to sell a Class I or Class II medical device need only submit a 510(k) pre-market notification. A clinical study must be done in accordance with an Investigational Drug Exemption (IDE) in support of a Pre-Market Approval application (PMA) for a Class III device, which is subject to the toughest regulation.
Additionally, in India, the Ministry of Health, in collaboration with the DCGI and the ICMR, developed Good Clinical Practises (GCP-India) standards as an ethical and scientific quality standard for the design and conduct of human subjects trials.
Fairfield’s Ranking Board
Top Segments
- Phase III Category Continues to Dominate
With a share of sales of 53.5% of the global market in 2025, the Phase III sector had the highest revenue. Phase III trials, which contain the largest subject populations and the highest costs, are the reason for this. Phase III also necessitates a larger patient base and frequently a longer duration of therapy.
Furthermore, the phase II category is projected to experience the fastest market growth. The two steps of this study are the first stage, which entails investigating a variety of doses and performing efficacy tests, and the second stage, which entails choosing the dose. Due to the several drugs and vaccines that are currently in Phase II and are suggested for the treatment of COVID-19, the segment will grow.
- Interventional Studies Category Spearheads
In 2025, the interventional studies dominated the industry. This method is one of the most widely used in clinical studies. As of May 2020, the bulk of these studies were interventional behavioral, clinical procedure, and device investigations, with the remainder being trialed for drugs or biologics. At that time, 79.0% of all registered studies were interventional studies.
The expanded access category is anticipated to grow substantially throughout the projected period. There may be a means for people with severe sickness problems to receive therapy outside of the trial if there are no appropriate medicines accessible. The extended access trials segment is expected to be driven by growing innovation in clinical trial methodologies.
- Oncology Remains the Leading Segment
The oncology segment led the market in 2025 because of its large revenue. The segment is expanding as a result of rising cancer treatment drug approval rates and rising need for efficient cancer medicines. Clinical development has mostly focused on non-small cell lung cancer (NSCLC) and breast cancer, among the several oncology indications.
The cardiovascular category is expected to experience the fastest growth within the forecast time frame. Due to the massive investment being made in R&D in this industry as a result of the expanding prevalence and global demand for cost-effective drugs, more than 190 pharmaceuticals are now being developed. It is anticipated that rising government R&D spending will increase demand for affordable pharmaceuticals in low- and middle-income nations, supporting market expansion.
- Demand for Clinical Trial Data Management Services Maximum
In 2025, the clinical trial data management services category led the market growth. The demand for clinical data management systems is anticipated to increase as there are more clinical trials conducted, particularly in the Asia Pacific area. The demand for quick and cutting-edge technologies to perform clinical trials is growing as the number of trials rises.
As a result, clinical data management tools are utilised in clinical research to manage such large amounts of data because they are one of the sponsors' most valuable assets. Moreover, the laboratory services category is expected to grow fastest in the clinical trials market during the forecast period.
The sector is growing as a result of factors like the growing cases of chronic diseases and the rising demand for early diagnostic tests. Additionally, it is projected that during the forecast period, quick developments in sample preparation and data management will propel sector growth.
Regional Frontrunners
The US Secures Leadership Position for North America
Because of factors like the pharmaceutical industry's high R&D spending, the presence of well-established players, the region's robust regulatory structure, the rising prevalence of diseases, and the sizeable contribution of the United States, the North American region is anticipated to contribute to the market growth during the study period significantly.
Additionally, the support provided by the governments of the region's nations is fostering market expansion. The participation of the region's top market players in the development of new drugs and devices is a second factor that is projected to boost market expansion in the area.
Asia Pacific Fastest on the Back of a Large Accessible Patient Pool for Clinical Trial Participation
Asia Pacific, on the other hand, is predicted to grow at the fastest pace during the forecast period. This is because a sizable patient pool is easily accessible, making it simpler to find participants for clinical trials. Due to their excellent quality and quick turnaround, "Novotech," Asia Pacific's largest and most seasoned biotech CRO, has seen a rise in requests for studies from biotechnology sponsors.
There is potential for the market to expand due to the region's growing need for people with obesity, cancer, and cardiovascular diseases. The rise in ageing population, and widespread age-related health difficulties are to blame for the market expansion in nations like China, and Japan. The market in this area is expanding at a faster rate thanks to rising government and private organisation funding for the cheap delivery of medications.
Fairfield’s Competitive Landscape Analysis
The global clinical trials market is a congested industry with few significant companies operating worldwide. To increase their global footprint, the major firms are launching new items and enhancing their distribution networks. In addition, Fairfield Market Research anticipates that during the next few years, there will be further market consolidation.
Who are the Leaders in the Global Clinical Trials Space?
- Eli Lilly and Company
- Syneos Health
- Pharmaceutical Product Development (PPD)
- Novo Nordisk A/S
- Icon plc.
- Charles River Laboratories
- Parexel International Corporation
- WuXi Apptec
- Medpace Holdings, Inc.
- KCR Pharmaceuticals Pvt. Ltd.
- Pharmaceutical Solutions Industry Ltd. (PSI).
- Pfizer
- Thermo Fisher Scientific Inc.
- Sanofi SA
Global Clinical Trials Market is Segmented as Below:
By Phase:
- Phase I
- Phase II
- Phase III
- Phase IV
By Study Design:
- Interventional
- Observational
- Expanded Access
By Indication:
- Autoimmune/Inflammation
- Rheumatoid arthritis
- Multiple Sclerosis
- Osteoarthritis
- Irritable Bowel Syndrome (IBS)
- Others
- Pain Management
- Chronic Pain
- Acute Pain
- Oncology
- Blood Cancer
- Solid Tumours
- Other
- CNS Condition
- Epilepsy
- Parkinson's Disease (PD)
- Huntington's Disease
- Stroke
- Traumatic Brain Injury (TBI)
- Amyotrophic Lateral Sclerosis (ALS)
- Muscle Regeneration
- Others
- Diabetes
- Obesity
- Cardiovascular
- Others
By Service Type:
- Protocol Designing
- Site Identification
- Patient Recruitment
- Laboratory Services
- Bioanalytical Testing Services
- Clinical Trial Data Management Services
- Others
By Geographic Coverage:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
- Executive Summary
- Global Clinical Trials Market Snapshot
- Future Projections
- Key Market Trends
- Regional Snapshot, by Value, 2026
- Analyst Recommendations
- Market Overview
- Market Definitions and Segmentations
- Market Dynamics
- Drivers
- Restraints
- Market Opportunities
- Value Chain Analysis
- COVID-19 Impact Analysis
- Porter's Five Forces Analysis
- Impact of Russia-Ukraine Conflict
- PESTLE Analysis
- Regulatory Analysis
- Price Trend Analysis
- Current Prices and Future Projections, 2025-2033
- Price Impact Factors
- Global Clinical Trials Market Outlook, 2020 - 2033
- Global Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Phase I
- Phase II
- Phase III
- Phase III
- Global Clinical Trials Market Outlook, by Study Design, Value (US$ Mn), 2020-2033
- Interventional
- Observational
- Expanded Access
- Global Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- Global Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- Global Clinical Trials Market Outlook, by Region, Value (US$ Mn), 2020-2033
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
- Global Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- North America Clinical Trials Market Outlook, 2020 - 2033
- North America Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Phase I
- Phase II
- Phase III
- Phase III
- North America Clinical Trials Market Outlook, by Study Design, Value (US$ Mn), 2020-2033
- Interventional
- Observational
- Expanded Access
- North America Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- North America Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- North America Clinical Trials Market Outlook, by Country, Value (US$ Mn), 2020-2033
- U.S. Clinical Trials Market Outlook, by Phase, 2020-2033
- U.S. Clinical Trials Market Outlook, by Study Design, 2020-2033
- U.S. Clinical Trials Market Outlook, by Indication, 2020-2033
- U.S. Clinical Trials Market Outlook, by Indication, 2020-2033
- Canada Clinical Trials Market Outlook, by Phase, 2020-2033
- Canada Clinical Trials Market Outlook, by Study Design, 2020-2033
- Canada Clinical Trials Market Outlook, by Indication, 2020-2033
- Canada Clinical Trials Market Outlook, by Indication, 2020-2033
- BPS Analysis/Market Attractiveness Analysis
- North America Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Europe Clinical Trials Market Outlook, 2020 - 2033
- Europe Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Phase I
- Phase II
- Phase III
- Phase III
- Europe Clinical Trials Market Outlook, by Study Design, Value (US$ Mn), 2020-2033
- Interventional
- Observational
- Expanded Access
- Europe Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- Europe Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- Europe Clinical Trials Market Outlook, by Country, Value (US$ Mn), 2020-2033
- Germany Clinical Trials Market Outlook, by Phase, 2020-2033
- Germany Clinical Trials Market Outlook, by Study Design, 2020-2033
- Germany Clinical Trials Market Outlook, by Indication, 2020-2033
- Germany Clinical Trials Market Outlook, by Indication, 2020-2033
- Italy Clinical Trials Market Outlook, by Phase, 2020-2033
- Italy Clinical Trials Market Outlook, by Study Design, 2020-2033
- Italy Clinical Trials Market Outlook, by Indication, 2020-2033
- Italy Clinical Trials Market Outlook, by Indication, 2020-2033
- France Clinical Trials Market Outlook, by Phase, 2020-2033
- France Clinical Trials Market Outlook, by Study Design, 2020-2033
- France Clinical Trials Market Outlook, by Indication, 2020-2033
- France Clinical Trials Market Outlook, by Indication, 2020-2033
- U.K. Clinical Trials Market Outlook, by Phase, 2020-2033
- U.K. Clinical Trials Market Outlook, by Study Design, 2020-2033
- U.K. Clinical Trials Market Outlook, by Indication, 2020-2033
- U.K. Clinical Trials Market Outlook, by Indication, 2020-2033
- Spain Clinical Trials Market Outlook, by Phase, 2020-2033
- Spain Clinical Trials Market Outlook, by Study Design, 2020-2033
- Spain Clinical Trials Market Outlook, by Indication, 2020-2033
- Spain Clinical Trials Market Outlook, by Indication, 2020-2033
- Russia Clinical Trials Market Outlook, by Phase, 2020-2033
- Russia Clinical Trials Market Outlook, by Study Design, 2020-2033
- Russia Clinical Trials Market Outlook, by Indication, 2020-2033
- Russia Clinical Trials Market Outlook, by Indication, 2020-2033
- Rest of Europe Clinical Trials Market Outlook, by Phase, 2020-2033
- Rest of Europe Clinical Trials Market Outlook, by Study Design, 2020-2033
- Rest of Europe Clinical Trials Market Outlook, by Indication, 2020-2033
- Rest of Europe Clinical Trials Market Outlook, by Indication, 2020-2033
- BPS Analysis/Market Attractiveness Analysis
- Europe Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Asia Pacific Clinical Trials Market Outlook, 2020 - 2033
- Asia Pacific Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Phase I
- Phase II
- Phase III
- Phase III
- Asia Pacific Clinical Trials Market Outlook, by Study Design, Value (US$ Mn), 2020-2033
- Interventional
- Observational
- Expanded Access
- Asia Pacific Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- Asia Pacific Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- Asia Pacific Clinical Trials Market Outlook, by Country, Value (US$ Mn), 2020-2033
- China Clinical Trials Market Outlook, by Phase, 2020-2033
- China Clinical Trials Market Outlook, by Study Design, 2020-2033
- China Clinical Trials Market Outlook, by Indication, 2020-2033
- China Clinical Trials Market Outlook, by Indication, 2020-2033
- Japan Clinical Trials Market Outlook, by Phase, 2020-2033
- Japan Clinical Trials Market Outlook, by Study Design, 2020-2033
- Japan Clinical Trials Market Outlook, by Indication, 2020-2033
- Japan Clinical Trials Market Outlook, by Indication, 2020-2033
- South Korea Clinical Trials Market Outlook, by Phase, 2020-2033
- South Korea Clinical Trials Market Outlook, by Study Design, 2020-2033
- South Korea Clinical Trials Market Outlook, by Indication, 2020-2033
- South Korea Clinical Trials Market Outlook, by Indication, 2020-2033
- India Clinical Trials Market Outlook, by Phase, 2020-2033
- India Clinical Trials Market Outlook, by Study Design, 2020-2033
- India Clinical Trials Market Outlook, by Indication, 2020-2033
- India Clinical Trials Market Outlook, by Indication, 2020-2033
- Southeast Asia Clinical Trials Market Outlook, by Phase, 2020-2033
- Southeast Asia Clinical Trials Market Outlook, by Study Design, 2020-2033
- Southeast Asia Clinical Trials Market Outlook, by Indication, 2020-2033
- Southeast Asia Clinical Trials Market Outlook, by Indication, 2020-2033
- Rest of SAO Clinical Trials Market Outlook, by Phase, 2020-2033
- Rest of SAO Clinical Trials Market Outlook, by Study Design, 2020-2033
- Rest of SAO Clinical Trials Market Outlook, by Indication, 2020-2033
- Rest of SAO Clinical Trials Market Outlook, by Indication, 2020-2033
- BPS Analysis/Market Attractiveness Analysis
- Asia Pacific Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Latin America Clinical Trials Market Outlook, 2020 - 2033
- Latin America Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Phase I
- Phase II
- Phase III
- Phase III
- Latin America Clinical Trials Market Outlook, by Study Design, Value (US$ Mn), 2020-2033
- Interventional
- Observational
- Expanded Access
- Latin America Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- Latin America Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- Latin America Clinical Trials Market Outlook, by Country, Value (US$ Mn), 2020-2033
- Brazil Clinical Trials Market Outlook, by Phase, 2020-2033
- Brazil Clinical Trials Market Outlook, by Study Design, 2020-2033
- Brazil Clinical Trials Market Outlook, by Indication, 2020-2033
- Brazil Clinical Trials Market Outlook, by Indication, 2020-2033
- Mexico Clinical Trials Market Outlook, by Phase, 2020-2033
- Mexico Clinical Trials Market Outlook, by Study Design, 2020-2033
- Mexico Clinical Trials Market Outlook, by Indication, 2020-2033
- Mexico Clinical Trials Market Outlook, by Indication, 2020-2033
- Argentina Clinical Trials Market Outlook, by Phase, 2020-2033
- Argentina Clinical Trials Market Outlook, by Study Design, 2020-2033
- Argentina Clinical Trials Market Outlook, by Indication, 2020-2033
- Argentina Clinical Trials Market Outlook, by Indication, 2020-2033
- Rest of LATAM Clinical Trials Market Outlook, by Phase, 2020-2033
- Rest of LATAM Clinical Trials Market Outlook, by Study Design, 2020-2033
- Rest of LATAM Clinical Trials Market Outlook, by Indication, 2020-2033
- Rest of LATAM Clinical Trials Market Outlook, by Indication, 2020-2033
- BPS Analysis/Market Attractiveness Analysis
- Latin America Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Middle East & Africa Clinical Trials Market Outlook, 2020 - 2033
- Middle East & Africa Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Phase I
- Phase II
- Phase III
- Phase III
- Middle East & Africa Clinical Trials Market Outlook, by Study Design, Value (US$ Mn), 2020-2033
- Interventional
- Observational
- Expanded Access
- Middle East & Africa Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- Middle East & Africa Clinical Trials Market Outlook, by Indication, Value (US$ Mn), 2020-2033
- Autoimmune/Inflammation
- Pain Management
- CNS Condition
- Diabetes
- Obesity
- Cardiovascular
- Others
- Middle East & Africa Clinical Trials Market Outlook, by Country, Value (US$ Mn), 2020-2033
- GCC Clinical Trials Market Outlook, by Phase, 2020-2033
- GCC Clinical Trials Market Outlook, by Study Design, 2020-2033
- GCC Clinical Trials Market Outlook, by Indication, 2020-2033
- GCC Clinical Trials Market Outlook, by Indication, 2020-2033
- South Africa Clinical Trials Market Outlook, by Phase, 2020-2033
- South Africa Clinical Trials Market Outlook, by Study Design, 2020-2033
- South Africa Clinical Trials Market Outlook, by Indication, 2020-2033
- South Africa Clinical Trials Market Outlook, by Indication, 2020-2033
- Egypt Clinical Trials Market Outlook, by Phase, 2020-2033
- Egypt Clinical Trials Market Outlook, by Study Design, 2020-2033
- Egypt Clinical Trials Market Outlook, by Indication, 2020-2033
- Egypt Clinical Trials Market Outlook, by Indication, 2020-2033
- Nigeria Clinical Trials Market Outlook, by Phase, 2020-2033
- Nigeria Clinical Trials Market Outlook, by Study Design, 2020-2033
- Nigeria Clinical Trials Market Outlook, by Indication, 2020-2033
- Nigeria Clinical Trials Market Outlook, by Indication, 2020-2033
- Rest of Middle East Clinical Trials Market Outlook, by Phase, 2020-2033
- Rest of Middle East Clinical Trials Market Outlook, by Study Design, 2020-2033
- Rest of Middle East Clinical Trials Market Outlook, by Indication, 2020-2033
- Rest of Middle East Clinical Trials Market Outlook, by Indication, 2020-2033
- BPS Analysis/Market Attractiveness Analysis
- Middle East & Africa Clinical Trials Market Outlook, by Phase, Value (US$ Mn), 2020-2033
- Competitive Landscape
- Company Vs Segment Heatmap
- Company Market Share Analysis, 2025
- Competitive Dashboard
- Company Profiles
- Eli Lilly and Company
- Company Overview
- Product Portfolio
- Financial Overview
- Business Strategies and Developments
- Syneos Health
- Pharmaceutical Product Development (PPD)
- Novo Nordisk A/S
- Icon plc.
- Charles River Laboratories
- Parexel International Corporation
- WuXi Apptec
- Medpace Holdings, Inc.
- KCR Pharmaceuticals Pvt. Ltd.
- Eli Lilly and Company
- Appendix
- Research Methodology
- Report Assumptions
- Acronyms and Abbreviations
|
BASE YEAR |
HISTORICAL DATA |
FORECAST PERIOD |
UNITS |
|||
|
2025 |
|
2019 - 2024 |
2026 - 2033 |
Value: US$ Billion |
||
|
REPORT FEATURES |
DETAILS |
|
Phase Coverage |
|
|
Study Design Coverage |
|
|
Indication Coverage |
|
|
Service Type Coverage |
|
|
Geographical Coverage |
|
|
Leading Companies |
|
|
Report Highlights |
Key Market Indicators, Macro-micro economic impact analysis, Technological Roadmap, Key Trends, Driver, Restraints, and Future Opportunities & Revenue Pockets, Porter’s 5 Forces Analysis, Historical Trend (2019-2021), Market Estimates and Forecast, Market Dynamics, Industry Trends, Competition Landscape, Category, Region, Country-wise Trends & Analysis, COVID-19 Impact Analysis (Demand and Supply Chain) |
