Global Fructosamine Test Market Growth and Industrial Forecast, 2019-2032
Executive Summary & Key Highlights
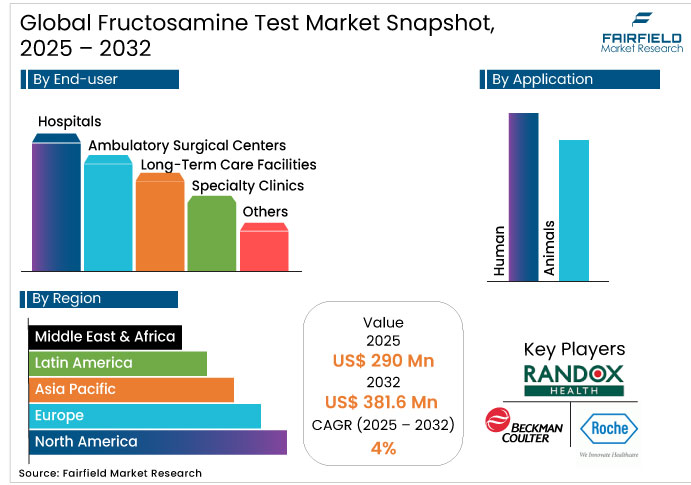
- The global fructosamine test market reached approximately US$290 million in 2025 and is projected to grow to US$381.6 million by 2032, reflecting a CAGR of about 4%. This indicates a mature market, supported by steady, reliable demand rather than rapid or disruptive growth.
- Type II Diabetes leads the Disease Indication segment with a dominant 55% share in 2025, driving strong fructosamine adoption across Hospitals and Specialty Clinics globally.
- Blood Serum remains the leading sample Type with 81% market share by 2025 due to established laboratory workflows across North America and Europe.
- Blood Plasma is the fastest-growing sample Type, expanding steadily through 2032 as distributed and Point-of-Care testing gains adoption in emerging Asia and Latin America.
- Clinical Diagnostics for Human applications dominate with 56% market value in 2025, driven by routine diabetes management in inpatient and outpatient settings.
- Clinical Diagnostics for Animals market growth is attributed to the overall market as companion animal diabetes and veterinary laboratory capabilities expanding in Western Europe and North America.
- Hospitals lead the End-User landscape with 30% share, reflecting high-volume automated testing and centralized procurement structures.
- Asia Pacific is the fastest-growing region, projected to contribute nearly 30% of global market growth in 2032, fueled by China’s 90M and India’s 70M diabetic populations.
Market Dynamics: Drivers, Restraints, and Opportunities Analysis
Market Drivers
- Technological Advancement in Laboratory Automation and Point-of-Care Testing
Technological innovation is reshaping the Fructosamine Test Market as automated immunoassay platforms and enzymatic assay methodologies enhance specificity, analytical throughput, and turnaround times versus colorimetric methods. The rise of portable point-of-care devices, supported by regulatory easing for POC approvals, is accelerating decentralized testing adoption. AI and machine learning integration is also improving diagnostic precision and clinician decision-making.
Siemens Healthcare's automated fructosamine assays for ADVIA Chemistry systems and Randox Laboratories’ enzymatic test kits reflect this technological progress. These advancements reduce manual intervention, boost analytical reliability, and improve EHR workflow integration across diabetes clinics and diagnostic centers. Sustained R&D investments continue to reinforce this structural driver within diabetes diagnostics innovation.
- Expansion of Healthcare Infrastructure and Diagnostic Testing Accessibility in Emerging Markets
Asia-Pacific fructosamine test markets are witnessing rapid healthcare investment and diagnostic expansion, especially in China and India. China’s fructosamine test market expanded significantly in 2025 and is projected to grow steadily, supported by rising diabetes incidence and strengthened laboratory infrastructure. India reflects similarly fast-growing dynamics driven by urbanization, lifestyle shifts, and government-backed healthcare access programs.
World Bank and OECD data show sustained healthcare spending increases across emerging economies, boosting diagnostic infrastructure and reshaping global competitiveness. Regional manufacturers are expanding production and distribution capabilities for both domestic and export markets, attracting companies deploying capital into emerging regions. However, success requires navigating diverse regulatory systems, reimbursement variations, and infrastructure development complexities.

Market Restraints
- Limited Clinical Awareness and Reimbursement Uncertainty
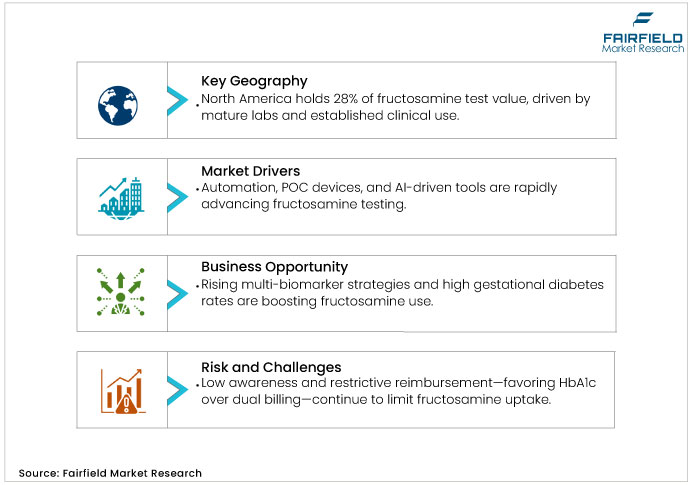
Fructosamine testing adoption remains limited due to low clinical awareness in certain segments and emerging markets. Reimbursement policies often prohibit concurrent billing of fructosamine and HbA1c, with Medicare and commercial insurers prioritizing HbA1c as the primary biomarker. This restricts testing volume in price-sensitive systems and limits provider incentives.
Reimbursement uncertainty is estimated to restrain fructosamine test market growth, with the strongest impact in North America and Western Europe where policies are most stringent. Diagnostic providers can mitigate this restraint through outcome-driven evidence and health economic studies. Such initiatives can support future reimbursement policy evolution and broader adoption.
- Competition from Alternative Glycemic Monitoring Methodologies
HbA1c remains the dominant standard for long-term glycemic monitoring, while CGM technologies are emerging with real-time glucose insights that appeal to specific patient groups. Additional alternatives such as OGTT, fasting plasma glucose, and hemoglobin-variant specific assays further fragment the testing landscape. This collective competition suppresses fructosamine adoption by reducing clinical reliance on multi-analyte strategies.
Fructosamine test market consolidation around high-volume modalities creates switching barriers that slow fructosamine uptake. Manufacturers can offset this challenge through clinical differentiation focused on scenarios such as hemoglobinopathy management, renal disease monitoring, and pregnancy-related glucose assessment. These areas highlight unique, documented advantages of fructosamine testing.
Market Opportunities
- Integration of Fructosamine Testing into Comprehensive Diabetes Management Protocols
Growing emphasis on multi-biomarker diabetes assessment is creating new opportunities for fructosamine integration within diagnostic algorithms. Gestational diabetes is a key underserved segment where fructosamine provides clear advantages, avoiding interference from hemoglobin variants or CKD. With gestational diabetes affecting roughly one in five pregnancies globally and showing 11–25% prevalence in select emerging regions, the addressable demand is significant.
International Diabetes Federation recommendations for gestational diabetes and early-onset Type I pediatric diabetes further expand targeted deployment potential. Realizing this opportunity requires guideline updates, workflow redesign for multi-analyte testing, and EHR integration to support clinical decision-making.
- Point-of-Care and Home-Based Fructosamine Testing Expansion
The shift toward decentralized care and patient-directed monitoring is opening strong opportunities for POC and home-based fructosamine testing. Current penetration is modest and largely limited to specialized diabetes clinics and research settings. User-friendly, rapid-turnaround POC platforms could expand adoption across primary care, remote care, and home environments, especially in emerging markets with limited lab infrastructure.
The addressable market value for POC fructosamine testing is expected to expand significantly through 2032 if adoption accelerates. Success depends on generating evidence of POC reliability comparable to central labs, securing FDA/CE approvals, and integrating workflows into healthcare systems. Companies achieving early entry into POC fructosamine platforms stand to capture substantial market share.
Regional Market Assessment: Strategic Geography Analysis
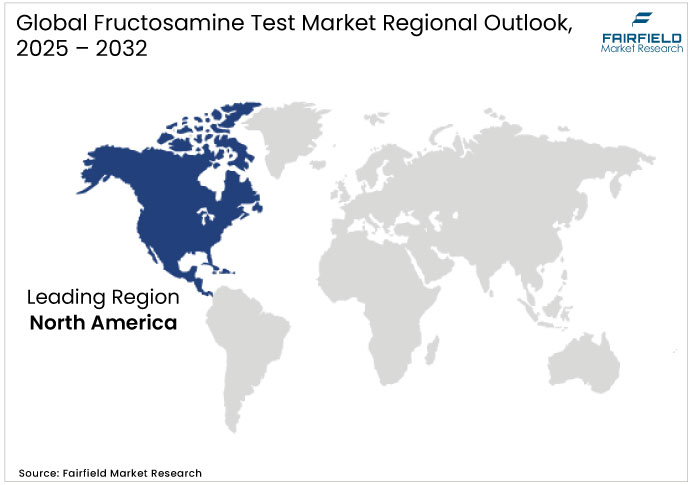
- North America: Mature Market with Technology Leadership
North America, led by the U.S., accounts for 28% of global fructosamine test market value in 2025, with the U.S. market growing at a modest single-digit CAGR through 2032. This maturity reflects advanced infrastructure, strong laboratory networks, and established clinical use in hemoglobin-related conditions and intensive insulin therapy. Reimbursement expansion continues slowly as supporting clinical evidence increases.
FDA and CLIA regulations shape a competitive environment centered on validation, quality, and performance standards. Innovation driven by Abbott and Siemens Healthineers advances immunoassay automation across major urban hubs. As healthcare consolidation centralizes procurement in large IDNs, differentiation increasingly depends on system integration and value-based service models rather than standalone product innovation.
- Europe: Regulatory Harmonization and Differentiated Market Dynamics
Europe shows wide variation in fructosamine test market dynamics, with Germany and the United Kingdom together representing 10–15% of regional fructosamine testing demand. CE marking and IVD Regulation 2017/746 create harmonized compliance standards, though reimbursement policies still vary nationally. Sustainability, data security, and advanced diagnostic adoption are increasingly shaping competitive positioning.
Growth is driven by expanding diabetes screening programs in Western and Nordic countries, alongside emerging adoption in Central and Eastern Europe. Successful expansion requires navigating country-specific reimbursement systems, procurement pathways, and clinical adoption timelines. Moderate long-term growth through 2032 is supported by healthcare investment and aging demographics, with differentiation tied to integration capabilities and IVD regulatory compliance.
- Asia-Pacific: Differential Growth and Competitive Positioning
Asia-Pacific is the fastest-growing region, projected to reach US$115 million by 2032 and account for nearly 30% of global fructosamine test market growth acceleration. China contributes 10–15% of regional value, supported by 90 million diabetics (2021) and major healthcare modernization. India is the fastest-growing market, with over 70 million diabetics (2021) and strong expansion driven by urbanization and rising incomes.
Regional manufacturing strength, supply chain depth, and innovation capability are reshaping global competitiveness. Investment strategies must account for country-specific growth rates, regulatory environments, healthcare maturity, and reimbursement complexity. Expanding diagnostic infrastructure, especially in emerging markets will sustain Asia-Pacific growth above global averages through 2032, benefiting manufacturers with strong distribution and localized development.
Segmentation Analysis: Category-Wise Strategic Assessment
- Disease Indication Segment Analysis
Type II diabetes holds 55% of the fructosamine test market share in 2025, driven by rising global incidence linked to obesity, sedentary lifestyles, and aging populations. Fructosamine is increasingly used when HbA1c is unreliable, such as in anemia or hemoglobinopathy. Providers also adopt it for patients requiring frequent monitoring or showing unstable glucose control.
Type I diabetes remains smaller but is growing as intensive insulin therapy requires rapid fructosamine-based assessment. Gestational diabetes is still underpenetrated despite strong clinical need, though protocol integration and evidence generation may expand adoption through 2032. Both segments show rising potential as guidelines emphasize more frequent glycemic monitoring.
- Sample Type Segment Analysis
Blood serum sampling leads the fructosamine test market with 80% share by 2025, supported by long-established clinical protocols, strong lab infrastructure compatibility, and proven performance in automated systems. Its dominance reflects organizational and technological lock-in across workflows and quality assurance. Manufacturers focus on improving enzymatic assay specificity, reducing turnaround times, and enhancing automation integration.
Blood plasma is an emerging alternative, growing at a sustainable CAGR through 2032, supported by evidence of comparable performance and advantages for certain patient groups. Plasma-based assays offer differentiation in outpatient and distributed testing where serum processing is limited. The segment shows steady technology-driven expansion and moderate adoption through the forecast period.
- Application Segment Analysis
Clinical diagnostics for human patients leads the market with 56% of global fructosamine test market value by 2025, driven by routine diabetes management across inpatient and outpatient settings. Growth depends on protocol adoption, clinical efficacy validation, and reimbursement support. Competitive dynamics center on provider relationships and navigation of healthcare payment systems.
Animal clinical diagnostics is expanding rapidly, due to rising companion animal diabetes testing and veterinary lab development. The segment remains underpenetrated but offers strong potential through 2032. Firms with veterinary-specific assay formats and reference lab distribution can capture above-market growth.
- End-User Segment Analysis
Hospitals lead the end-user landscape with 30% of the fructosamine test market value in 2025, driven by centralized procurement, automated testing infrastructure, and integrated diabetes protocols. High-throughput hospital laboratories concentrate testing volumes within major central lab networks. This structure favors manufacturers offering automation-ready platforms and volume-based commercial models.
Specialty clinics are a fast-growing segment as endocrinology, metabolic centers, and diabetes clinics expand fructosamine use for treatment optimization. Ambulatory surgical centers and long-term care facilities show emerging adoption for perioperative and geriatric glucose monitoring. The fragmented multi-end-user mix requires tailored commercial strategies aligned with varied workflows and procurement patterns.
Competitive Landscape: Market Structure and Strategic Positioning
The fructosamine test market is moderately fragmented, with multinational diagnostics leaders viz., Roche, Abbott, and Siemens Healthineers—dominating central laboratory platform integration. These companies leverage automation platforms, healthcare system relationships, and broad diagnostic portfolios for bundled offerings. Specialized reagent and consumable manufacturers support point-of-care and niche applications within this structure.
Entry barriers remain high due to FDA 510(k) and CE marking requirements, evidence-generation costs, procurement relationships, and manufacturing quality demands. While POC development requires lower capital than central lab automation, allowing smaller players to enter, consolidation continues through M&A as larger corporations acquire specialized assay developers and regional distributors to expand capability and presence.
Key Players
- Hoffmann-La Roche Ltd
- Beckman Coulter, Inc. (Danaher)
- Randox Laboratories Ltd.
- Fortress Diagnostics
- KAMIYA BIOMEDICAL COMPANY
- Abbexa Ltd.
- Eurolyser Diagnostica GmbH
- Asahi Kasei Pharma Corporation
- Diazyme Laboratories
- Abcam plc
- BioVision, Inc.
- MyBioSource, Inc.
- Siemens Healthineers
- SENTINEL CH. SpA
- Weldon Biotech, Inc.
- Others
Global Fructosamine Test Market Segmentation-
By Disease Indication
- Diabetes I
- Diabetes II
- Gestational Diabetes
By Sample
- Blood Serum
- Blood Plasma
By Application
- Human
- Animals
By End-user
- Hospitals
- Ambulatory Surgical Centers
- Long-Term Care Facilities
- Specialty Clinics
- Others
By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
1. Executive Summary
1.1. Global Fructosamine Test Market Snapshot
1.2. Future Projections
1.3. Key Market Trends
1.4. Regional Snapshot, by Value, 2025
1.5. Analyst Recommendations
2. Market Overview
2.1. Market Definitions and Segmentations
2.2. Market Dynamics
2.2.1. Drivers
2.2.2. Restraints
2.2.3. Market Opportunities
2.3. Value Chain Analysis
2.4. COVID-19 Impact Analysis
2.5. Porter's Fiver Forces Analysis
2.6. Impact of Russia-Ukraine Conflict
2.7. PESTLE Analysis
2.8. Regulatory Analysis
2.9. Price Trend Analysis
2.9.1. Current Prices and Future Projections, 2024-2032
2.9.2. Price Impact Factors
3. Global Fructosamine Test Market Outlook, 2019 - 2032
3.1. Global Fructosamine Test Market Outlook, by Disease Indication, Value (US$ Bn), 2019-2032
3.1.1. Diabetes I
3.1.2. Diabetes II
3.1.3. Gestational Diabetes
3.2. Global Fructosamine Test Market Outlook, by Sample, Value (US$ Bn), 2019-2032
3.2.1. Blood Serum
3.2.2. Blood Plasma
3.3. Global Fructosamine Test Market Outlook, by Application, Value (US$ Bn), 2019-2032
3.3.1. Human
3.3.2. Animals
3.4. Global Fructosamine Test Market Outlook, by End-user, Value (US$ Bn), 2019-2032
3.4.1. Hospitals
3.4.2. Ambulatory Surgical Centers
3.4.3. Long-Term Care Facilities
3.4.4. Specialty Clinics
3.4.5. Others
3.5. Global Fructosamine Test Market Outlook, by Region, Value (US$ Bn), 2019-2032
3.5.1. North America
3.5.2. Europe
3.5.3. Asia Pacific
3.5.4. Latin America
3.5.5. Middle East & Africa
4. North America Fructosamine Test Market Outlook, 2019 - 2032
4.1. North America Fructosamine Test Market Outlook, by Disease Indication, Value (US$ Bn), 2019-2032
4.1.1. Diabetes I
4.1.2. Diabetes II
4.1.3. Gestational Diabetes
4.2. North America Fructosamine Test Market Outlook, by Sample, Value (US$ Bn), 2019-2032
4.2.1. Blood Serum
4.2.2. Blood Plasma
4.3. North America Fructosamine Test Market Outlook, by Application, Value (US$ Bn), 2019-2032
4.3.1. Human
4.3.2. Animals
4.4. North America Fructosamine Test Market Outlook, by End-user, Value (US$ Bn), 2019-2032
4.4.1. Hospitals
4.4.2. Ambulatory Surgical Centers
4.4.3. Long-Term Care Facilities
4.4.4. Specialty Clinics
4.4.5. Others
4.5. North America Fructosamine Test Market Outlook, by Country, Value (US$ Bn), 2019-2032
4.5.1. U.S. Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
4.5.2. U.S. Fructosamine Test Market Outlook, by Sample, 2019-2032
4.5.3. U.S. Fructosamine Test Market Outlook, by Application, 2019-2032
4.5.4. U.S. Fructosamine Test Market Outlook, by End-user, 2019-2032
4.5.5. Canada Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
4.5.6. Canada Fructosamine Test Market Outlook, by Sample, 2019-2032
4.5.7. Canada Fructosamine Test Market Outlook, by Application, 2019-2032
4.5.8. Canada Fructosamine Test Market Outlook, by End-user, 2019-2032
4.6. BPS Analysis/Market Attractiveness Analysis
5. Europe Fructosamine Test Market Outlook, 2019 - 2032
5.1. Europe Fructosamine Test Market Outlook, by Disease Indication, Value (US$ Bn), 2019-2032
5.1.1. Diabetes I
5.1.2. Diabetes II
5.1.3. Gestational Diabetes
5.2. Europe Fructosamine Test Market Outlook, by Sample, Value (US$ Bn), 2019-2032
5.2.1. Blood Serum
5.2.2. Blood Plasma
5.3. Europe Fructosamine Test Market Outlook, by Application, Value (US$ Bn), 2019-2032
5.3.1. Human
5.3.2. Animals
5.4. Europe Fructosamine Test Market Outlook, by End-user, Value (US$ Bn), 2019-2032
5.4.1. Hospitals
5.4.2. Ambulatory Surgical Centers
5.4.3. Long-Term Care Facilities
5.4.4. Specialty Clinics
5.4.5. Others
5.5. Europe Fructosamine Test Market Outlook, by Country, Value (US$ Bn), 2019-2032
5.5.1. Germany Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
5.5.2. Germany Fructosamine Test Market Outlook, by Sample, 2019-2032
5.5.3. Germany Fructosamine Test Market Outlook, by Application, 2019-2032
5.5.4. Germany Fructosamine Test Market Outlook, by End-user, 2019-2032
5.5.5. Italy Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
5.5.6. Italy Fructosamine Test Market Outlook, by Sample, 2019-2032
5.5.7. Italy Fructosamine Test Market Outlook, by Application, 2019-2032
5.5.8. Italy Fructosamine Test Market Outlook, by End-user, 2019-2032
5.5.9. France Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
5.5.10. France Fructosamine Test Market Outlook, by Sample, 2019-2032
5.5.11. France Fructosamine Test Market Outlook, by Application, 2019-2032
5.5.12. France Fructosamine Test Market Outlook, by End-user, 2019-2032
5.5.13. U.K. Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
5.5.14. U.K. Fructosamine Test Market Outlook, by Sample, 2019-2032
5.5.15. U.K. Fructosamine Test Market Outlook, by Application, 2019-2032
5.5.16. U.K. Fructosamine Test Market Outlook, by End-user, 2019-2032
5.5.17. Spain Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
5.5.18. Spain Fructosamine Test Market Outlook, by Sample, 2019-2032
5.5.19. Spain Fructosamine Test Market Outlook, by Application, 2019-2032
5.5.20. Spain Fructosamine Test Market Outlook, by End-user, 2019-2032
5.5.21. Russia Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
5.5.22. Russia Fructosamine Test Market Outlook, by Sample, 2019-2032
5.5.23. Russia Fructosamine Test Market Outlook, by Application, 2019-2032
5.5.24. Russia Fructosamine Test Market Outlook, by End-user, 2019-2032
5.5.25. Rest of Europe Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
5.5.26. Rest of Europe Fructosamine Test Market Outlook, by Sample, 2019-2032
5.5.27. Rest of Europe Fructosamine Test Market Outlook, by Application, 2019-2032
5.5.28. Rest of Europe Fructosamine Test Market Outlook, by End-user, 2019-2032
5.6. BPS Analysis/Market Attractiveness Analysis
6. Asia Pacific Fructosamine Test Market Outlook, 2019 - 2032
6.1. Asia Pacific Fructosamine Test Market Outlook, by Disease Indication, Value (US$ Bn), 2019-2032
6.1.1. Diabetes I
6.1.2. Diabetes II
6.1.3. Gestational Diabetes
6.2. Asia Pacific Fructosamine Test Market Outlook, by Sample, Value (US$ Bn), 2019-2032
6.2.1. Blood Serum
6.2.2. Blood Plasma
6.3. Asia Pacific Fructosamine Test Market Outlook, by Application, Value (US$ Bn), 2019-2032
6.3.1. Human
6.3.2. Animals
6.4. Asia Pacific Fructosamine Test Market Outlook, by End-user, Value (US$ Bn), 2019-2032
6.4.1. Hospitals
6.4.2. Ambulatory Surgical Centers
6.4.3. Long-Term Care Facilities
6.4.4. Specialty Clinics
6.4.5. Others
6.5. Asia Pacific Fructosamine Test Market Outlook, by Country, Value (US$ Bn), 2019-2032
6.5.1. China Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
6.5.2. China Fructosamine Test Market Outlook, by Sample, 2019-2032
6.5.3. China Fructosamine Test Market Outlook, by Application, 2019-2032
6.5.4. China Fructosamine Test Market Outlook, by End-user, 2019-2032
6.5.5. Japan Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
6.5.6. Japan Fructosamine Test Market Outlook, by Sample, 2019-2032
6.5.7. Japan Fructosamine Test Market Outlook, by Application, 2019-2032
6.5.8. Japan Fructosamine Test Market Outlook, by End-user, 2019-2032
6.5.9. South Korea Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
6.5.10. South Korea Fructosamine Test Market Outlook, by Sample, 2019-2032
6.5.11. South Korea Fructosamine Test Market Outlook, by Application, 2019-2032
6.5.12. South Korea Fructosamine Test Market Outlook, by End-user, 2019-2032
6.5.13. India Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
6.5.14. India Fructosamine Test Market Outlook, by Sample, 2019-2032
6.5.15. India Fructosamine Test Market Outlook, by Application, 2019-2032
6.5.16. India Fructosamine Test Market Outlook, by End-user, 2019-2032
6.5.17. Southeast Asia Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
6.5.18. Southeast Asia Fructosamine Test Market Outlook, by Sample, 2019-2032
6.5.19. Southeast Asia Fructosamine Test Market Outlook, by Application, 2019-2032
6.5.20. Southeast Asia Fructosamine Test Market Outlook, by End-user, 2019-2032
6.5.21. Rest of SAO Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
6.5.22. Rest of SAO Fructosamine Test Market Outlook, by Sample, 2019-2032
6.5.23. Rest of SAO Fructosamine Test Market Outlook, by Application, 2019-2032
6.5.24. Rest of SAO Fructosamine Test Market Outlook, by End-user, 2019-2032
6.6. BPS Analysis/Market Attractiveness Analysis
7. Latin America Fructosamine Test Market Outlook, 2019 - 2032
7.1. Latin America Fructosamine Test Market Outlook, by Disease Indication, Value (US$ Bn), 2019-2032
7.1.1. Diabetes I
7.1.2. Diabetes II
7.1.3. Gestational Diabetes
7.2. Latin America Fructosamine Test Market Outlook, by Sample, Value (US$ Bn), 2019-2032
7.2.1. Blood Serum
7.2.2. Blood Plasma
7.3. Latin America Fructosamine Test Market Outlook, by Application, Value (US$ Bn), 2019-2032
7.3.1. Human
7.3.2. Animals
7.4. Latin America Fructosamine Test Market Outlook, by End-user, Value (US$ Bn), 2019-2032
7.4.1. Hospitals
7.4.2. Ambulatory Surgical Centers
7.4.3. Long-Term Care Facilities
7.4.4. Specialty Clinics
7.4.5. Others
7.5. Latin America Fructosamine Test Market Outlook, by Country, Value (US$ Bn), 2019-2032
7.5.1. Brazil Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
7.5.2. Brazil Fructosamine Test Market Outlook, by Sample, 2019-2032
7.5.3. Brazil Fructosamine Test Market Outlook, by Application, 2019-2032
7.5.4. Brazil Fructosamine Test Market Outlook, by End-user, 2019-2032
7.5.5. Mexico Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
7.5.6. Mexico Fructosamine Test Market Outlook, by Sample, 2019-2032
7.5.7. Mexico Fructosamine Test Market Outlook, by Application, 2019-2032
7.5.8. Mexico Fructosamine Test Market Outlook, by End-user, 2019-2032
7.5.9. Argentina Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
7.5.10. Argentina Fructosamine Test Market Outlook, by Sample, 2019-2032
7.5.11. Argentina Fructosamine Test Market Outlook, by Application, 2019-2032
7.5.12. Argentina Fructosamine Test Market Outlook, by End-user, 2019-2032
7.5.13. Rest of LATAM Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
7.5.14. Rest of LATAM Fructosamine Test Market Outlook, by Sample, 2019-2032
7.5.15. Rest of LATAM Fructosamine Test Market Outlook, by Application, 2019-2032
7.5.16. Rest of LATAM Fructosamine Test Market Outlook, by End-user, 2019-2032
7.6. BPS Analysis/Market Attractiveness Analysis
8. Middle East & Africa Fructosamine Test Market Outlook, 2019 - 2032
8.1. Middle East & Africa Fructosamine Test Market Outlook, by Disease Indication, Value (US$ Bn), 2019-2032
8.1.1. Diabetes I
8.1.2. Diabetes II
8.1.3. Gestational Diabetes
8.2. Middle East & Africa Fructosamine Test Market Outlook, by Sample, Value (US$ Bn), 2019-2032
8.2.1. Blood Serum
8.2.2. Blood Plasma
8.3. Middle East & Africa Fructosamine Test Market Outlook, by Application, Value (US$ Bn), 2019-2032
8.3.1. Human
8.3.2. Animals
8.4. Middle East & Africa Fructosamine Test Market Outlook, by End-user, Value (US$ Bn), 2019-2032
8.4.1. Hospitals
8.4.2. Ambulatory Surgical Centers
8.4.3. Long-Term Care Facilities
8.4.4. Specialty Clinics
8.4.5. Others
8.5. Middle East & Africa Fructosamine Test Market Outlook, by Country, Value (US$ Bn), 2019-2032
8.5.1. GCC Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
8.5.2. GCC Fructosamine Test Market Outlook, by Sample, 2019-2032
8.5.3. GCC Fructosamine Test Market Outlook, by Application, 2019-2032
8.5.4. GCC Fructosamine Test Market Outlook, by End-user, 2019-2032
8.5.5. South Africa Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
8.5.6. South Africa Fructosamine Test Market Outlook, by Sample, 2019-2032
8.5.7. South Africa Fructosamine Test Market Outlook, by Application, 2019-2032
8.5.8. South Africa Fructosamine Test Market Outlook, by End-user, 2019-2032
8.5.9. Egypt Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
8.5.10. Egypt Fructosamine Test Market Outlook, by Sample, 2019-2032
8.5.11. Egypt Fructosamine Test Market Outlook, by Application, 2019-2032
8.5.12. Egypt Fructosamine Test Market Outlook, by End-user, 2019-2032
8.5.13. Nigeria Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
8.5.14. Nigeria Fructosamine Test Market Outlook, by Sample, 2019-2032
8.5.15. Nigeria Fructosamine Test Market Outlook, by Application, 2019-2032
8.5.16. Nigeria Fructosamine Test Market Outlook, by End-user, 2019-2032
8.5.17. Rest of Middle East Fructosamine Test Market Outlook, by Disease Indication, 2019-2032
8.5.18. Rest of Middle East Fructosamine Test Market Outlook, by Sample, 2019-2032
8.5.19. Rest of Middle East Fructosamine Test Market Outlook, by Application, 2019-2032
8.5.20. Rest of Middle East Fructosamine Test Market Outlook, by End-user, 2019-2032
8.6. BPS Analysis/Market Attractiveness Analysis
9. Competitive Landscape
9.1. Company Vs Segment Heatmap
9.2. Company Market Share Analysis, 2024
9.3. Competitive Dashboard
9.4. Company Profiles
9.4.1. F. Hoffmann-La Roche Ltd
9.4.1.1. Company Overview
9.4.1.2. Product Portfolio
9.4.1.3. Financial Overview
9.4.1.4. Business Strategies and Developments
9.4.2. Beckman Coulter, Inc. (Danaher)
9.4.2.1. Company Overview
9.4.2.2. Product Portfolio
9.4.2.3. Financial Overview
9.4.2.4. Business Strategies and Developments
9.4.3. Randox Laboratories Ltd.
9.4.3.1. Company Overview
9.4.3.2. Product Portfolio
9.4.3.3. Financial Overview
9.4.3.4. Business Strategies and Developments
9.4.4. Fortress Diagnostics
9.4.4.1. Company Overview
9.4.4.2. Product Portfolio
9.4.4.3. Financial Overview
9.4.4.4. Business Strategies and Developments
9.4.5. KAMIYA BIOMEDICAL COMPANY
9.4.5.1. Company Overview
9.4.5.2. Product Portfolio
9.4.5.3. Financial Overview
9.4.5.4. Business Strategies and Developments
9.4.6. Abbexa Ltd.
9.4.6.1. Company Overview
9.4.6.2. Product Portfolio
9.4.6.3. Financial Overview
9.4.6.4. Business Strategies and Developments
9.4.7. Eurolyser Diagnostica GmbH
9.4.7.1. Company Overview
9.4.7.2. Product Portfolio
9.4.7.3. Financial Overview
9.4.7.4. Business Strategies and Developments
9.4.8. Asahi Kasei Pharma Corporation
9.4.8.1. Company Overview
9.4.8.2. Product Portfolio
9.4.8.3. Financial Overview
9.4.8.4. Business Strategies and Developments
9.4.9. Diazyme Laboratories
9.4.9.1. Company Overview
9.4.9.2. Product Portfolio
9.4.9.3. Financial Overview
9.4.9.4. Business Strategies and Developments
9.4.10. Abcam plc
9.4.10.1. Company Overview
9.4.10.2. Product Portfolio
9.4.10.3. Financial Overview
9.4.10.4. Business Strategies and Developments
10. Appendix
10.1. Research Methodology
10.2. Report Assumptions
10.3. Acronyms and Abbreviations
|
BASE YEAR |
HISTORICAL DATA |
FORECAST PERIOD |
UNITS |
|||
|
2024 |
|
2019 - 2024 |
2025 - 2032 |
Value: US$ Billion |
||
|
REPORT FEATURES |
DETAILS |
|
By Disease Indication Coverage |
|
|
By Sample Coverage |
|
|
By Application Coverage |
|
|
By End-User Coverage |
|
|
Geographical Coverage |
|
|
Leading Companies |
|
|
Report Highlights |
Key Market Indicators, Macro-micro economic impact analysis, Technological Roadmap, Key Trends, Driver, Restraints, and Future Opportunities & Revenue Pockets, Porter’s 5 Forces Analysis, Historical Trend (2019-2024), Market Estimates and Forecast, Market Dynamics, Industry Trends, Competition Landscape, Category, Region, Country-wise Trends & Analysis, COVID-19 Impact Analysis (Demand and Supply Chain) |
